Context Is Everything: The Role of the Microenvironment in Regulation of Gene Expression
Given that all cells in an organism have the same genetic material, the Bissell laboratory wants to learn the basis of organ and tissue-specificity, i.e. how different organs such as the mammary gland (also referred to as breast in humans) ‘know’ to be normal and what goes wrong when they turn into breast cancer. Mina Bissell was one of the first to propose and elucidate the important signaling role of the extracellular-matrix (ECM)-the large molecules that are outside the cells and provide many functions for the cells and organs in these processes. She recognized the crucial role of the microenvironment surrounding each tissue and organ and of the three-dimensional (3D) tissue structure in differentiation and cancer as factors that determined tissue-specificity. Together with her collaborators, the Bissell laboratory has developed unique and versatile 3D organotypic models that can be used to rapidly distinguish normal and malignant cells. These techniques (or assays) now are much appreciated and used widely in basic biological research as well as drug testing.
In the last 3 decades, through an imaginative blend of cell and molecular biology, the Bissell laboratory has utilized these models to support the theory of “dynamic reciprocity” proposed by Bissell which was published in 1982 (Bissell et al, J. Theoretical Biology); namely, that the ECM exerts physical and biochemical influences on a cell, which are then transduced by cell surface receptors through the cell’s skeleton to ultimately effect changes in gene expression within the nucleus. In turn the cell, in this now altered state, can remodel the ECM (for instance, by inducing expression of matrix metalloproteinases, the enzymes that digest the ECM). Not only does this feedback to the cell nucleus (remember, this is a dynamic and reciprocal interaction), but the ripple effect throughout the ECM can also affect other cells near and far. Currently, work in the Bissell laboratory is focused on answering questions related to each level of these microenvironmental interactions: from the development of complex Microenvironment Array (ME ArrayTM) platforms to determine how combinations of different ECM proteins and soluble signaling molecules direct cell behavior and stem cell fate; to identifying cytoskeletal factors that influence tissue architecture and conduct transfer of signals from the outside to the nucleus to identifying how changes in the ECM signaling alter tissue-specificity and influence the expression of key genes.
Finally since the mammary gland, like all organs, is composed of multiple cell types, and since the laboratory is interested in understanding not only ‘cell biology’ but also tissue, organ and cancer biology, we continue to engineer culture platforms to determine how tissue architecture and other cell types influence normal development and tumor progression.
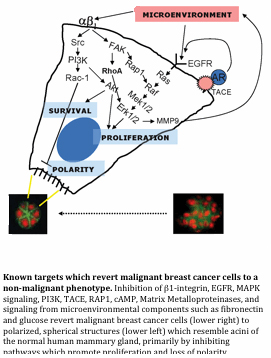 We have been able to determine a number of factors that are key to inducing normal function in mammary epithelial cells, and how some of these signals go awry during tumor development. It is clear that the large cross-shaped ECM protein, laminin-111 (LN1), which is an important component of the basement membrane that surrounds the basal surface of the mammary epithelium, is key to proper organization, and architecture as well as to eliciting proper gene expression such as the expression and secretion of milk proteins. These cells line the ducts that secrete and harbor milk within the breast. Furthermore, these milk-producing epithelial cells are the root of breast carcinoma, and we have also discovered (with our long-time collaborator, Dr. Ole Petersen of the University of Copenhagen) that the muscle-like myoepithelial cells that surround the ducts and squeeze out milk also play this crucial role in vivo: they secrete LN1 to orient and confer function upon luminal epithelial cells so that these cells can become polar, and secrete milk and other proteins in the proper directions. Myoepithelial cells are known to be lost as breast cancer progresses, and the concomitant loss of LN1 causes luminal cells to disorganize, impacting the intricate architecture of the breast. The loss of this important signal nudges luminal epithelial cells towards a disorganized state. We have shown that such aberrant signaling can cause genomic instability and ultimately result in malignancy. We have been able to determine a number of factors that are key to inducing normal function in mammary epithelial cells, and how some of these signals go awry during tumor development. It is clear that the large cross-shaped ECM protein, laminin-111 (LN1), which is an important component of the basement membrane that surrounds the basal surface of the mammary epithelium, is key to proper organization, and architecture as well as to eliciting proper gene expression such as the expression and secretion of milk proteins. These cells line the ducts that secrete and harbor milk within the breast. Furthermore, these milk-producing epithelial cells are the root of breast carcinoma, and we have also discovered (with our long-time collaborator, Dr. Ole Petersen of the University of Copenhagen) that the muscle-like myoepithelial cells that surround the ducts and squeeze out milk also play this crucial role in vivo: they secrete LN1 to orient and confer function upon luminal epithelial cells so that these cells can become polar, and secrete milk and other proteins in the proper directions. Myoepithelial cells are known to be lost as breast cancer progresses, and the concomitant loss of LN1 causes luminal cells to disorganize, impacting the intricate architecture of the breast. The loss of this important signal nudges luminal epithelial cells towards a disorganized state. We have shown that such aberrant signaling can cause genomic instability and ultimately result in malignancy.
These results pointed us also to the opposite question, i.e. if aberrant signaling can make cells and tissues disorganized and eventually malignant, could we in turn make breast cancer cells think they are ‘normal’ by giving them signals that could replace those provided by myoepithelial cells to restore their architecture and halt their growth? Astonishingly, we have demonstrated that breast carcinoma cells harboring many genetic alterations can still ‘revert’ to a normal phenotype in 3D cell culture by correcting aberrant signaling from the extracellular environment to reestablish proper tissue polarity. These findings could shed light on why tumor cells become dormant and what could ‘awaken’ them to become malignant again. We also have used the ‘reversion assay’ to develop a functional screen and have discovered new signaling pathways that play key roles in keeping cells functional.
For more technical data and details of these experiments, we invite you to browse our list of publications for articles related to these and other findings.
|

