The role of two closely related proteins, long
suspected of being major contributors to the development of a number of
cancers, has at last been identified. Researchers with the U.S. Department
of Energy's Lawrence Berkeley National Laboratory (Berkeley Lab) have
shown that the proteins known as "Ski" and "Sno" block
the "downstream" events initiated by TGF-ß (transforming growth
factor-beta), an extracellular protein responsible for controlling the
growth and differentiation of certain types of cells.
 |

CELL BIOLOGIST KUNXIN LUO LED THE THE TEAM THAT DISCOVERED HOW
SKI AND SNO CONTRIBUTE TO THE DEVELOPMENT OF CANCER
|
The TGF-ß protein sends a signal that stops the growth of epithelial
cells – the cells that line the skin, kidney, glands, lungs,
gastrointestinal tract, bladder, and blood vessels. If something goes
awry, causing the TGF-ß signal to be blocked, cell growth continues
unchecked, giving rise to cancerous tumors. Nearly 90-percent of all human
cancers involve epithelial cells.
A research team led by Kunxin Luo, a cell biologist with Berkeley Lab's
Life Sciences Division, and an assistant adjunct professor with the
University of California at Berkeley, has been able to demonstrate that
Ski and Sno proteins interact with a family of proteins called "Smad"
to completely shut down the TGF-ß signal. Smad proteins are known tumor
suppressors.
"We've shown that Ski and Sno, two oncogene products, directly
interact with the protein products of tumor suppressor genes at a common
point," says Luo. "This indicates that oncoproteins and tumor
suppressor proteins do not act independently. Rather, they regulate one
another's function. Our work also suggests that cancer development is a
delicate balancing act in which the balance gets tipped the wrong
way."
 |
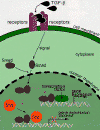
CLICK TO SEE ILLUSTRATION OF HOW SKI & SNO REGULATE CELL
GROWTH PATHWAYS
|
Although the way in which Ski interacts with Smad proteins to control
TGF-ß signals continues to be a mystery, Luo and her colleagues have
reported that the mechanism behind the effects of Sno is a negative
feedback loop (see Science, October 22, 1999).
TGF-ß proteins cannot enter cells and must therefore transmit their
signals by attaching themselves to receptor proteins on a cell's outer
surface. The signal generated by this interaction is then carried across
the cell membrane and into the nucleus via Smad proteins.
Luo and her colleagues discovered that a normal level of Sno inside the
nucleus blunts TGF-ß signals, but as the number of Smad proteins
increases, the level of Sno drops until it is low enough for the signals
to take effect.
"After two hours, the TGF-ß signals have resulted in a marked
increase in the expression of the Sno gene," says Luo. "The
level of Sno rises until it blocks the functions of Smad. This allows a
cell to resume normal growth activity."
When Luo and her colleagues deliberately kept levels of Ski and Sno
artificially high, the TGF-ß signal was blocked and cell growth could no
longer be restrained. It is unclear at this time to what degree Ski and
Sno act independently or in conjunction with one another, and what factors
cause the balancing act between these two proteins and the Smad proteins
to be tipped in a cancerous direction.
"Cancer is not a simple disease and there are many different
pathways through which it can develop," says Luo. "However,
knowing the relevant levels of Ski and Sno in a cell gives us a possible
tool for early cancer diagnosis."
Luo and her colleagues were able to identify the roles of Ski and Sno
in cancer development by working with liver cancer cells of a type known
as Hep3B cells. They were looking to identify proteins in the cell nucleus
that interacted with the Smad proteins which earlier work had identified
as carriers of the TGF-ß signal. To do this, they engineered a Smad
protein to host a special molecular "tag" that would be
recognized by an antibody. Tagged Smad proteins could then be introduced
into cells and subsequently harvested using the antibody. Caught up in the
harvest would be any other type of protein that might be attached to the
tagged Smads.
"It was an emotional roller coaster ride of anticipation to see
what we would find when we analyzed our results," says Luo. "We
were quite surprised and very excited when we learned that we had captured
oncoproteins."
Specifically in their tests, Luo and her colleagues found Ski and SnoN,
the most predominant form of the Sno protein. In the immediate future, Luo
plans to investigate the ways in which oncoproteins interact with tumor
suppressors to regulate cell growth. This work has implications beyond
carcinogenesis.
For example, TGF-ß signals stop the growth of epithelial cells but
promote the growth of fibroblast cells. Says Luo, "The discovery of
Ski and Sno as blockers of the TGF-ß signal may lead to their
applications in the treatment of fibrosis diseases."
TGF-ß signals also promote the differentiation of other types of
cells, especially muscle cells. Studies have shown that Ski and Sno affect
this process as well – an excess of either results in an abundance of
muscle, a deficit yields the reverse. Other important functions of TGF-ß
signals include the healing of wounds and the repair of damaged tissue.
Members of the Luo group that worked on the Ski and SnoN oncoproteins
were Shannon Stroschein and Wei Wang. Their collaborators were Sharleen
Zhou and Qian Zhou from UC Berkeley. Also participating in the Ski
experiments were Dan Chen and Eric Martens from UC Berkeley.
Berkeley Lab is a U.S. Department of Energy national laboratory located
in Berkeley, California. It conducts unclassified scientific research and
is managed by the University of California.
|