Tapping into Solar Energy Riches: Berkeley Lab’s Helios Project and the Solar Energy Research Center
By Lynn Yarris
As stars go, from the standpoint of the universe, our Sun is nothing extraordinary – a yellow dwarf made up mostly of hydrogen just like at least 100 million other class G2 stars in the Milky Way. But for Earth, the Sun is a one-of-a-kind superstar – a bountiful source of energy that separates our bright living world from a dark dead rock. This same life-sustaining bounty of energy also represents humankind’s ultimate hope for developing energy technologies that are environmentally clean, carbon neutral – meaning they do not exacerbate global warming - and renewable for as long as it matters.
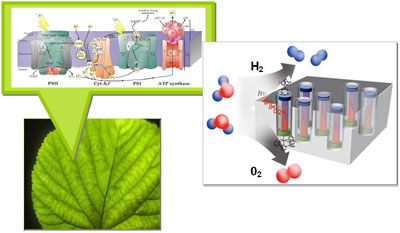
Within the leaves of green plants are photosynthetic (PS) reaction centers, protein complexes that capture light and use the energy to create and transport electrons that, with the help of a catalyst, split water molecules into oxygen molecules and hydrogen ions. Electrons are later used to reduce carbon dioxide for the formation of sugars. Scientists would like to create a synthetic leaf out of a membrane embedded with PEC cells. A water-oxidizing catalyst is attached to the anodic end of the PEC cell (red) and oxygen forms there when water molecules are split. Meanwhile, hydrogen ions are transported through the membrane and another type of catalyst at the cathodic end (blue) of the PEC cell aids in the formation of hydrogen molecules. Hydrogen, the simplest form of stored chemical energy, would then serve as a stepping stone for the production of liquid transportation fuels. (Image courtesy of Helios SERC)
How bountiful is the energy we get from the sun? Each year, the earth receives about 100,000 terawatts (trillion watts) of solar energy. With annual worldwide human energy consumption running at about 13 terawatts, this translates to enough power in an hour’s worth of global sunlight to meet an entire year’s worth of human energy needs. There is an enormous effort underway by scientists all over the world to develop various technologies that can tap into solar energy. At Berkeley Lab, one such effort is being carried out at the Solar Energy Research Center (SERC) under the project dubbed “Helios,” after the ancient Greek god who wore the sun as his crown and carried it across the sky in a fiery chariot. The goal at Helios SERC is to convert solar energy into electricity and then use that electricity to drive chemical reactions that convert water and carbon dioxide into liquid transportation fuel. In other words, Helios SERC researchers want to develop an artificial version of photosynthesis.
“Through photosynthesis, green plants collect sunlight and use it to produce energy in the form of sugars from water and carbon dioxide. This works very well for plants but the overall power efficiency is not high enough to meet the needs of a modern society,” says Paul Alivisatos, a chemist who serves as interim director of Berkeley Lab and director of Helios SERC, and who is one of the world’s leading authorities on nanoscience and its application to photovoltaics – solar cells that convert sunlight directly into electricity. “While we have the technology now to create artificial photosynthesis systems with adequate power efficiency, such systems are much too expensive and complex to use on a commercial scale.”
Researchers at Helios SERC are working towards artificial photosynthesis via several avenues of development – nanocrystals of photovoltaic materials that can serve as efficient solar light collectors and electrodes; nanocrystal catalysts that can initiate energy efficient chemistry for splitting water molecules and reducing carbon dioxide; and specialized membranes for embedding these nanocrystals so that the functions of light harvesting, electrical charge separation, and fuel production can be integrated into a single system.
Says Alivisatos, “In effect, by placing all the photosynthetic elements of light collection, electrical charge separation, and chemical catalysis to make fuels from water and carbon dioxide on the same membrane of photovoltaic material, we want to create a synthetic leaf."

Paul Alivisatos is the interim Director of Berkeley Lab and the Larry and Diane Bock professor of Nanotechnology at UC Berkeley. He is also an award-winning chemist recognized as one of the world’s leading authorities on the fabrication of nanocrystals and their application to solar energy.
This synthetic leaf would come in the form of a single, solar-powered chemical reactor called a PhotoElectroChemical (PEC) cell. Creating carbon-neutral liquid transportation fuels using PEC cells has the potential to be far more efficient than creating such fuels from biomass. However, the highest efficiency prototype PEC cells today are made from relatively scarce and expensive semiconductors such as large crystals of crystalline silicon, or thin films of cadmium telluride or copper indium gallium selenide. Alivisatos and his colleagues want to replace these semiconductors with novel photovoltaic materials that would make commercial-scale applications of PEC cells practical. They also want to synthesize new catalysts that are less expensive but more effective than what is available today.
“Our ultimate goal is to deploy an artificial photosynthetic system across a large geographical area, at a level of efficiency that could provide the United States a significant alternative fuel source,” says Alivisatos. “For example, if we were to cover 58 million acres of non-arable land at various locations with photovoltaic nanocrystals that operate at a conversion efficiency of even one-percent, we could generate enough energy to replace all gasoline consumption in the U.S. If we could reach a conversion efficiency of 10-percent we would far exceed the total energy consumption in this country today.”
Progress is being made. In a recent study of potential new photovoltaic materials undertaken by Alivisatos with Cyrus Wadia, a member of his research group, and Daniel Kammen, director of UC Berkeley’s Renewable and Appropriate Energy Laboratory, iron pyrite, or fool's gold, a mineral that is abundant and relatively cheap, was identified as having great potential for use in solar cells. In a Helios SERC study of potential catalysts, Heinz Frei, a chemist with Berkeley Lab’s Physical Biosciences Division, and his postdoctoral student Feng Jiao, discovered that nanocrystals of cobalt oxide can effectively carry out the critical photosynthetic reaction of splitting water molecules.
"This is one of the most exciting times in memory for energy research," Alivisatos says. “National laboratories, such as Berkeley Lab, are going to play an incredibly important role as the nation enters into a mode of going after the energy problem in new ways. The national labs can act as anchor points, working with universities and companies, so that basic and applied research interactions can take place in a timely way that will help the nation quickly move forward on energy. We're in a hurry."
Additional Information
- For more information about Helios SERC visit the Website at http://www.lbl.gov/LBL-Programs/helios-serc/index.html


