Goals & Challenges (page 1)
The goal of Helios SERC is to produce carbon-neutral transportation fuels using solar energy as the source of stored energy
SERC pursues a route that doesn’t include biological photosynthesis or biomass. Instead, SERC is involved in using sunlight to drive chemical reactions that can reform the atoms in water and carbon dioxide into liquid transportation fuels. This route does not depend on arable land, but does depend on a significant amount of captured sunlight and carbon dioxide.
To meet this challenging goal, scientists in SERC are developing an artificial photosynthesis process in which advanced materials and new molecules are used to accomplish a series of processing steps that are inspired by natural photosynthesis. In an artificial photosynthesis process, light harvesters (photovoltaic semiconductor elements (PVs) or light-absorbing molecules) collect sunlight and create electrical charges that are used to drive fuel-forming chemical reactions. These chemical reactions involve catalytic molecules that enable the efficient chemical conversion of carbon dioxide and water molecules into a liquid fuel that can be stored, transported, and used for transportation or other applications.
The effort in SERC is focused on building a highly affordable system in which the entire process will take place in a single photoelectrochemical, or PEC, cell that collects sunlight and then generates the fuels at catalysts powered by the light harvesting and charge separating units. With its 26 senior scientists working on this problem, SERC brings to this effort a world-class level of expertise in materials, catalysis, photochemistry and electrochemistry. Specialists in each of these areas span the approaches of synthesis, diagnostics, and computational investigations.
By bypassing the production of complex plant material, prototype PEC cells have been shown to exhibit sunlight to chemical fuel conversion efficiencies not attainable by the natural photosynthetic process. The challenge is to develop PEC cells that use materials that are rugged, abundant, inexpensive, non-toxic, and can be made on a very large scale. This challenge is made easier by the use of nanomaterials that can be synthesized, shaped and functionalized with photoactive components using new techniques, in a manner that “designs” the needed properties into materials which, though abundant, otherwise would not be good performers in PEC systems. LBNL and UC Berkeley scientists have been leaders in developing these nanomaterials and expertise, and in uncovering the design principles of natural photosynthesis.
An example of SERC’s effort is to develop a novel PEC cell in which tiny nanoparticle-sized photovoltaics (nano-PVs) are directly connected to individual catalytic molecules. These nanoscale elements are placed with defined orientation into a porous membrane. The assembled membrane becomes an artificial photosynthetic system that accomplishes separation of the fuel molecules from evolving oxygen, with a geometry resembling that of the biological photosynthetic apparatus found in a leaf.
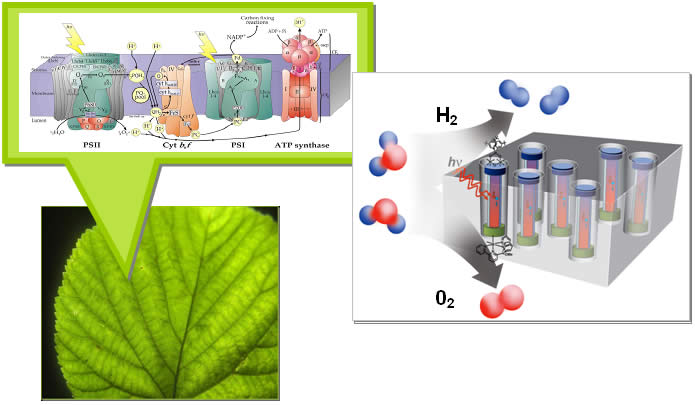
Above is a naturally occurring photosynthetic (PS) reaction center, as might be found in a leaf (figure courtesy Prof. J. Barber, ICL). Part of its complex mechanism, PSII in the figure, captures light, creates and transports electrons and positive charges that, with the help of a catalyst, form oxygen molecules and hydrogen ions from the oxidation of water. In later stages of the process, electrons are used to reduce carbon dioxide and “fix” the carbon in the formation of sugars.
The image on the right is an artist’s conceptual drawing of one of the membrane-based assemblies of oriented PEC cells that SERC is working to develop. Note that its geometry is similar to that of the natural photosynthetic system. The red/blue tubes are the embedded nanoPV’s. A catalyst that oxidizes water is attached to the anodic end (red) and oxygen is formed there from the oxidation of water molecules. The hydrogen ions are transported through the membrane and another type of catalyst at the cathodic end aids in the formation of hydrogen molecules. Hydrogen is the simplest form of stored chemical energy, and could be a stepping stone in the development of energy-rich fuels.
Progress requires overcoming major scientific CHALLENGES in the design of the photoactive and catalytic components
Research in SERC focuses on the breaking down of scientific barriers that have prevented so far the PV elements or light absorbing molecules, the fuel-forming catalysts, and the electrochemical elements to be sufficiently efficient, durable, and manufacturable from abundant materials using scalable processes.
CHALLENGE: Development of new light absorbing and charge-creating molecules and nanoPVs
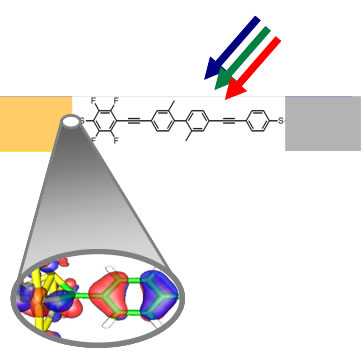
One type of hybrid nano PV element that is being explored in SERC consists of an organic macromolecule that absorbs sunlight and spontaneously separates charges. The contacts of the organic molecule with the metal electrode or catalyst surface (beige and gray squares) are stable chemical bonds made with atomic precision (shown in detail in the blown-up section). This quality of this contact assures that the separated charges are transported to and from the electrode or catalyst efficiently.
Scientific breakthroughs are needed to make PV elements that withstand the corrosive environment of photochemical water oxidation in aqueous media. We are opting for designs on the nanometer scale because electronic properties of semiconductor materials critical for efficient use of solar light can best be manipulated on this very small length scale. In fact, some of the most promising materials, especially alloys, exist only on the nanoscale. Moreover, nanoscale materials with extreme aspect ratios such as nanorods or nanotubes can be made that afford simultaneous optimization of efficiencies for the absorption of light and for transport of charges to the catalysts, both critical for achieving the required level of performance.
Another serious performance limitation of PV elements is poor contacts to catalysts or electrode surfaces. SERC researchers explore inorganic/organic hybrid PV elements with atomically designed covalent linkages of light absorbing polymer molecules and metal surface, a step that promises improved charge transport efficiency and durability. The effort involves the development of new synthetic methods and new techniques for atomic resolution manipulation, imaging and spectroscopic interrogation of nanoscale objects.



