|
YANO/YACHANDRA/KERN LAB :: RESEARCH LAWRENCE BERKELEY NATIONAL LABORATORY | MOLECULAR BIOPHYSICS AND INTEGRATED BIOIMAGING DIVISION |
|
|
home | [research] | people | photos | publications | contact | links |
|
|
OVERVIEWAlthough the global demand for energy is steadily increasing, most of the current sources of energy are either nonrenewable, nonsustainable, or contribute toward greenhouse gases in the environment. In addition to wind power and biofuels, solar energy is a renewable and clean alternative energy source, and development of methods to convert it into transportable fuels in a cost effective and efficient manner is a critical need. Solar energy is used efficiently in nature via the process of photosynthesis in plants, cyanobacteria, and algae. Studying how nature accomplishes these difficult reactions in natural photosynthetic systems and along with the study of artificial photosynthetic systems and other energy-relevant metalloenzymes is our overall research objective.
|

|
|
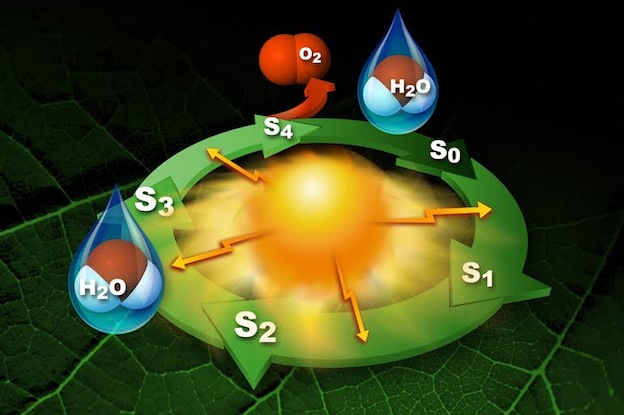
PHOTOSYSTEM II - WATER OXIDATION and O2 EVOLUTIONPhotosynthesis is a biochemical process by which green plants convert solar energy to carbohydrates. In this process, water is oxidized and dioxygen is formed via the photo-induced reaction:
The photosynthetic splitting of water (i.e. oxygen evolution) is the source of nearly all of the O2 in the atmosphere, and takes place in the oxygen evolving complex (OEC), which is located in the multisubunit membrane protein complex Photosystem II (PSII). The OEC is a cluster of four Mn atoms and one Ca atom (Mn4CaO5), and has been shown via x-ray spectroscopy to be linked by mono- and di-μ-oxo or hydroxo bridges. The OEC cycles through five different oxidation states, known as Si states (i = 0 - 4), coupling the one-electron photochemistry of the reaction center with the four-electron chemistry of water oxidation. Although the structure and mechanism of Mn4CaO5 has been extensively studied by various methods, the precise molecular details OF mechanism remain elusive.
|
|
|
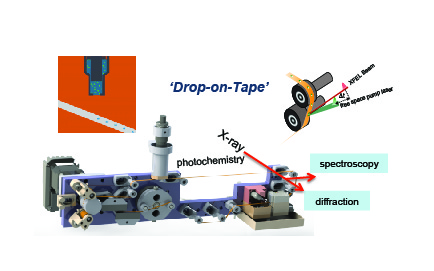
X-RAY FREE ELECTRON LASER (XFEL) STUDIESWe are using the femtosecond X-ray Free Electron Laser (XFEL) Facilities at Stanford, South Korea, Switzerland, and Japan to collect X-ray diffraction and emission spectra simultaneously. Our novel sample delivery method, shown on the right, facilitates room temperature, time dependent data collection. The droplet on tape method (DOT) utilizes an acoustic droplet dispensor to create droplets of crystal slurries onto a kapton tape. The tape travels through our micro environment before traveling to the XFEL beam. The micro environment can include laser illumination, gas saturation, or humidity changes. The droplet is timed with the pulse of the XFEL. Therefore, this method minimizes waste and maximizes micro environment felxibility. For more information on this method, please check out Fuller et al 2017, Young et al 2016, Jan et al 2018 and Ibrahim et al 2020.
|
|
|
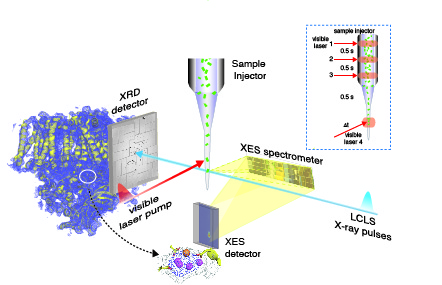
We utilize our delivery system at XFEL facilities to study biological systems. This unique method aids in simultaneous collection of X-ray diffraction (along the beam path) and spectroscopy (perpendicular to the beam). By collecting both data, we are able to correlate crystal structure with electronic information. By utilizing the high flux, time resolved nature of the XFEL, we can measure data from our biological samples without damage seen at traditional synchrotron facilities. Our research focuses on metalloenzymes, such as PS II described here.
|
|
|
X-RAY SPECTROSCOPY and X-RAY CRYSTALLOGRAPHY of PHOTOSYSTEM IIIn our ongoing investigation of electronic and geometric structure and changes of the Mn4CaO5 cluster, we have made extensive use of X-ray and EPR spectroscopy and X-ray crystallography. We are using high-resolution absorption and emission X-ray spectroscopy, including Resonant Inelastic X-ray Scattering spectroscopy, to study crystals and solutions of PS II and inorganic models. We use X-ray absorption spectra at the K-and L-edges of the Mn cluster in its native and intermediates states at room temperature in a time-resolved manner, to capture short-lived intermediates and the step that includes the O-O bond formation. These techniques that we are developing, using both XFELs and synchrotron sources, will elucidate not only the structure of the Mn4CaO5 catalytic complex, but also the changes in the structure as it cycles through the intermediate states, leading to an understanding of the mechanism. The insights gained from natural photosynthesis can then be applied in the design of inorganic-based energy conversion systems that split water, and will contribute to our search for cleaner, renewable carbon-neutral energy sources.
|
|
|
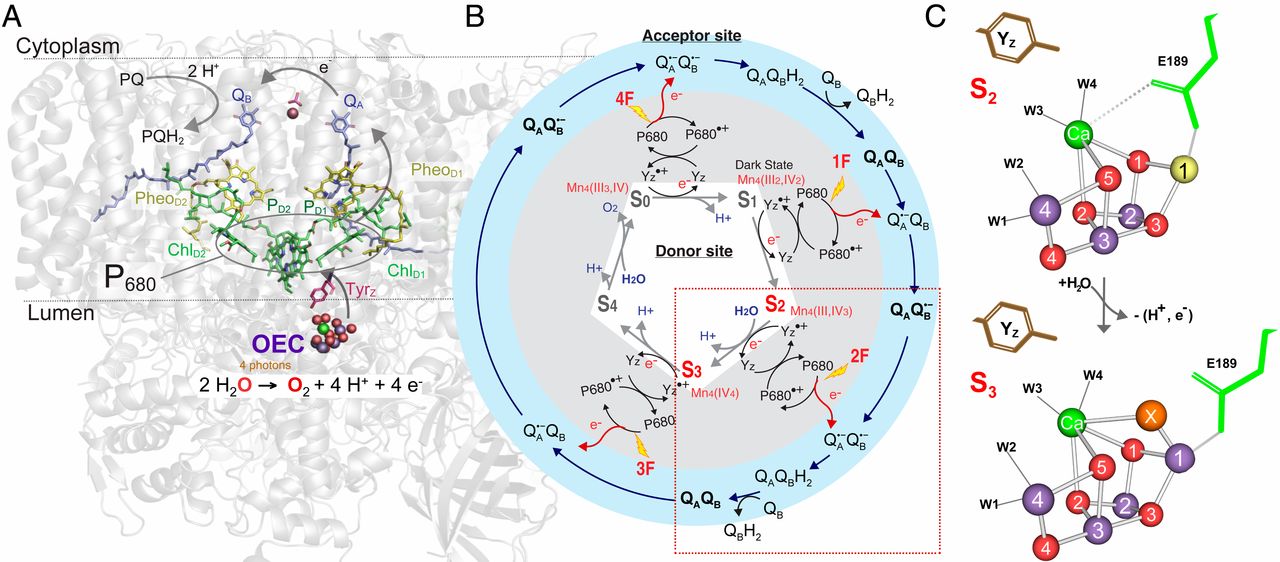
We are using XFELs and synchrotrons to systematically resolve the steps in the Kok cycle. Above is an image from our recent publication, 'Untangling the sequence of events during the S2 → S3 transition in photosytem II and implications for the water oxidation mechanism'. This paper focuses on the S2 → S3 transition. Utilizing the time dependence of our delivery method and the XFEL, we were able to resolve not only the stable intermediates (S2 and S3) but also time points between the S-states. Previously, we had seen the insertion of a new oxygen atom bridge between the Ca and Mn1 in the S3 state. By utilizing distinct time points, we are able to see when exactly this insertion happens. The goal is to collect data from many time points to make a molecular movie of the entire Kok cycle.
|
|
INORGANIC SYSTEMS FOR ENERGY CONVERSIONThe decades-long search for efficient, long-lived photo- and electrocatalysts for water oxidation, using earth-abundant and cost effective metals such as Mn, Fe, Co, Ni or others, has begun to produce important results. However, crucial breakthroughs are still required for the development of highly active, stable catalysts that promote rapid water splitting at a negligible overpotential. Significant improvements in catalyst performance have been hindered by a lack of structural and mechanistic understanding that will assist in the design of new catalytic structures.Promising catalytic materials include first row transition metal complexes. Related systems of importance are heterodinuclear units that exhibit tunability of redox properties. XAS/EXAFS/XES are ideal methods to determine the electronic and geometric structure of such materials. We have gained experience in developing and using such methods in our work with the natural photosynthetic system and we have begun to specifically tailor the methods for the study of artificial systems..
Although, steady state X-ray K- and L-absorption and emission techniques have been an enormously important tool for understanding the mechanism of these catalysts, it is critically important to be able to follow the reactions, in real time and in situ, to understand the dynamics of the systems. We are using in situ X-ray spectroscopy to study water-oxidation/CO2 reduction catalysts that are initiated chemically, photochemically or electrochemically, to understand the fundamental processes by which the catalysts operate and the phenomena that limit their effectiveness, so one can improve the efficiencies. We also developed methods for detecting the emission spectra of two elements from dilute systems simultaneously (Figure on the right) using an energy-dispersive emission spectrometer that can collect the entire emission spectra. This detection method allows us to follow the sequential chemistry at two metal sites following ultrafast laser pump and X-ray excitation. These proposed development of X-ray spectroscopy techniques for studying inorganic systems is in line with DOE interest in generating efficient catalysts for artificial photosynthetic systems.
|
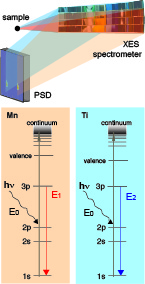 Simultaneous collection of the X-ray emission spectra from two transition metals.
|
|
STUDIES OF METALLOENZYMES RELEVANT TO ENERGYMetalloenzymes use metal ions in their active site to catalyze chemical transformation in a highly efficient manner. In order to achieve this unique capability, metalloproteins have evolved to maintain strict control over both the primary and secondary coordination spheres surrounding metal ions within active sites. They allow for fine tuning of the reactivity of the metal site. Thus, it is important to understand how the protein environment influences the chemistry at metal catalytic site by a combination of crystallography and X-ray spectroscopy. We are targeting transition metal based metalloenzymes and related systems to study the effect of the microenvironments that enables complicated chemistry in a sequential manner. Using simultaneous data collection of crystallography and X-ray spectroscopy at XFELs we study the interplay of metal sites and protein environment at room temperature. Some of the systems we have studied are methane monooxygenases (MMO), isopenicillin N synthase (IPNS), and ribonucleotide reductase (RNR), and other metalloenzymes related to energy are also being studied.
|
| home | [research] | people | photos | publications | contact | links |