Masters of atomic modeling
|
|
|
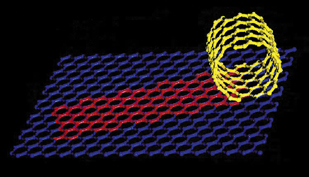
| A carbon nanotube is like a cylinder rolled up from a single sheet of graphite, whose atoms are arranged in hexagons. Depending on their "rolling" angle, nanotubes with different spirals have different electronic properties. | |
Marvin Cohen came to Berkeley from the University of Chicago in 1964, by way of Bell Telephone Laboratories. Louie was Cohen's student in the 1970s, then worked at IBM, Bell Laboratories, and the University of Pennsylvania before returning to Berkeley in 1980. They are masters of creating atom-by-atom models from "first principles," often using supercomputers at the National Energy Research Scientific Computing Center (NERSC).
The pseudopotential approach is a fundamental method to which Cohen has made crucial contributions since the 1960s. It views materials as an arrangement of charged atomic cores-nuclei and their "inert" inner electrons-immersed in a sea of valence electrons. The interaction among valence electrons can be modeled using Density Functional Theory for bonding and structural properties.
"To investigate a material all you need are the atomic weights and numbers," says Cohen. "The pseudopotential gives you valence electron-core interactions, and if you know the distribution of valence electron densities, you know what an electron will feel." Thus the theorist can put atoms in different arrangements and predict the material's energies-and because the lowest-energy arrangement is the most stable, "the lowest energy wins. You can map surfaces, find out where impurities will go, tell whether it will be superconducting, even create materials never seen before."
|
|
 |
 |
|
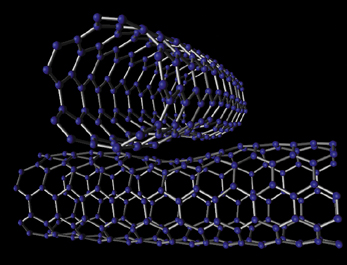
| (top) One way to deform nanotubes is to cross them. Because their electrical properties change as they are deformed, crossed-nanotube devices (bottom) may be used as ultrasensitive pressure switches. | |
Spectroscopic properties such as those measured at the Advanced Light Source (ALS) can also be predicted from first principles using many-body quantum theories, a field Louie pioneered in the 1980's.
After several years in which Cohen and Louie pursued their interests separately, a mutual fascination with nanotubes stimulated their collaboration.
The realm of the electron
"Because nanotubes are so small," says Louie, "the wavelength
of electrons is comparable to the dimension in which they are confined,"
and the electronic effects can be startling. Carbon in bulk in the form
of graphite is a semimetal, for example, but a carbon nanotube may be
a semiconductor or a metal "depending on how it's twisted."
As in a sheet of graphite, the atoms in a carbon nanotube are arranged in hexagons. If this atomic chicken wire is rolled so that the rows of eight hexagons go straight around the tube with their edges parallel to the axis, the electrons are forced to occupy fully separate energy bands. This kind of nanotube cannot conduct electricity unless the electrons get an energy boost across the gap. But if the chicken wire is rolled on the bias, electrons may not be confined to fully occupied energy bands, and the material may be a freely conducting metal.
Although electrons move according to the rules of quantum theory, there's a simple rule of thumb that predicts whether a chiral carbon nanotube will be a metal. "If you splice positions five steps across the grid and two steps up, for example, the difference is three, and it's a metal," Cohen says. "But it's a metal only if the difference is divisible by three-a twist that involves six steps across and two up, say, would give you another semiconductor."
To their own devices
|
|
 |
| Steven Louie and Marvin Cohen delight in creating atom-by-atom models, whether with sticks and balls or the world's most powerful supercomputers. | |
When Cohen and Louie found that nanotubes of different chirality could be joined in a bent junction by introducing five-member and seven-member rings, the potential for electronic devices was immediately obvious. Louie explains, "A metal joined to a semiconductor forms a basic device known as a Schottky diode"-ubiquitous in high-frequency, low-noise circuitry.
Another odd device which might be formed from carbon nanotubes, Cohen and Louie predicted, was an insulating junction of two metals. Normally a metal-to-metal junction would be conducting, but nanostructures are no bigger than the electron's wavelength. "A straight junction of two different metal nanotubes often would throw the electron out of phase, and it would stop," Louie says. A bent joint, on the other hand, should conduct.
It takes much microscopic sifting and poking about in sooty mats of carbon to find joined nanotubes, no matter what their chirality, so Louie- "frustrated by the difficulty of finding junctions"-has done theoretical studies on the simpler case of nanotubes laid across each other. His predictions are born out in crossed-tube systems built by Alex Zettl and Paul McEuen, experimenters with whom Cohen and Louie have worked closely for years-devices with electronics potential of their own.
Out of the matrix
Cohen, meanwhile, has concocted a truly futuristic nanogadget. Back-of-the-envelope
calculations indicated that "a cube just a hundredth of a centimeter
on a side could contain all the information in all the books ever printed.
Four wine bottles filled with nanotubes could match all the human brains
in the world." But how could one ever assemble the myriad elements
of a nanotube computer in a coherent fashion?
"I had a wild idea. Why not take mats of tubes as they come? Stick a thousand wires into a one-centimeter cube, randomly, feed in impulses, see what comes out-map what it can do, and then write software to use that." One could "lobotomize" useless or harmful structures, Cohen suggests, and even program the cube to "figure itself out. You leave it running at night, come back in the morning, and it's smarter."
Just how wild is this idea? When Cohen discussed it with executives at a major computer manufacturer recently, one of them remembered a long-ago experiment in which computers deliberately built with bad chips ran software designed to work around random hardware defects. "They're planning to run the experiment again, this time on molecular computers."
Unlimited potential
When it comes to making nanotubes, carbon is not the only game in town.
Cohen and Louie proposed nanotubes made of boron nitride, which act as
semiconductors no matter which way the chicken wire is wrapped; Zettl
soon synthesized them. They have designed tubes of boron, carbon, and
nitrogen, along which electrons would spiral from carbon to carbon, and
of boron carbide, a semiconductor that would form a metal when the tubes
are packed together in bundles.
|
|
|
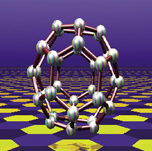
| Cohen and Louie predicted the existence of never-before-seen carbon-36 fullerences and characterized their remarkable properties, inspiring their experimental colleagues to synthesize them. | |
Nanotubes have extraordinarily high aspect ratios (length to diameter of millions to one) and, because of the extremely short bonds between atoms, they are the strongest material known- lengthwise. Rugged flat screens, atomic-scale "hypodermic needles," and solid storage tanks for hydrogen are among myriad applications under development.
And graphite-like bonds form other nanostructures as well. Nanococoons, enclosing nanocrystals of a different material, have already been made, as has carbon 36, crystals of small, rugby-shaped buckyballs with the potential to be superconducting at high temperature, which Cohen and Louie characterized before Zettl synthesized it. Nanopeapods, buckyballs nesting in tubes, have also recently been synthesized, and nano- "peashooters" that propel buckyballs may not be far behind.
While only part of the limitless universe of materials that have kept Marvin Cohen and Steven Louie and their postdocs and graduate students fascinated, nanostructure theory alone has generated enough predictions to keep experimenters busy for years.
SMARTY-PANTS MATERIALS
Sometimes a nanostructure can be
theorized down to the last electron.
At other times, the little devil surprises you.
Researchers in the Materials Sciences Division have lots of practice tailoring
materials that can respond to changes in acidity or temperature, or recognize
other substances like pathogens such as cholera or flu virus-often by
dramatically changing color or electrical conductivity.
Many of these so-called "smart materials" assemble themselves, making use of the special properties of surfactants-surface-active molecules. Stringy molecules called lipids have both hydrophilic (water-loving) and hydrophobic (water-fearing) ends, so they are called amphiphilic (amphi means both). When linked with like ends facing the same way, they can organize themselves into thin films, cylindrical structures, hollow spheres called liposomes, and other shapes.
Recently the group studying biosensors worked with lipids that bear the jaw-breaking moniker "bolaamphiphilic." These have both hydrophilic and hydrophobic ends, but their tails are tied together and there's a chemical group on each head, like rocks on a gaucho's bola.
Depending on the different chemical natures of their strings and head groups, molecular bolas display remarkable variety. One polymer membrane flaunts a kaleidoscopic array of shape changes in response to changes in acidity, forming ribbons that roll into spirals that tighten into tubes, which tighten still further into nanowires and frayed tangles. It's a wonderful repertoire of potentially beneficial behaviors-but nobody saw it coming.
Although many of the new lipid's properties were expected, says postdoctoral fellow Jie Song, some were surprising. "We knew the material would rigidify. But although we anticipated splitting in response to increased pH, the effect was far more dramatic than we had expected."
The sum of factors that underlie a promising new method of designing nanodevices makes fertile ground for investigation. - end -
| < Research Review | Top ^ | Next > |