The Garden of Converging Paths
 |
|
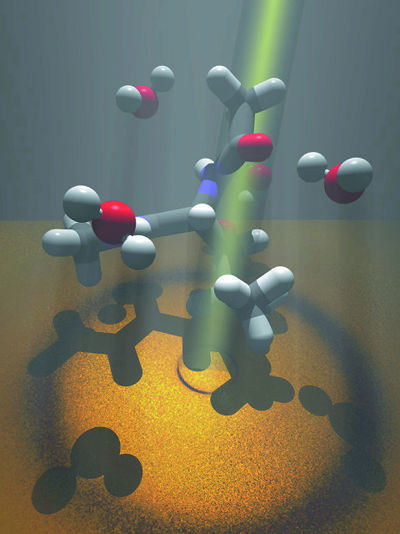
| To examine the structure of water near dissolved amino acids, Teresa Head-Gordon and her colleagues shine beams of x-rays or neutrons through leucine- the gray and white structures in this impressionistic illustration-dissolved in water, here represented by red and white structures. | |
One way to test ideas about how proteins fold is to start with a shape smaller and less intricate than most proteins, made from units less complicated than amino acids. Supercomputers simulate the behavior of model polymers, which in their native structure-analogous to the thermodynamically stable conformation of a fully folded protein-resemble jungle gyms made from Tinker-Toy-like sticks and balls.
Instead of the varying angles between amino-acid residues in a real protein, the stick-and-ball units, or mers, in a lattice model bond to their neighbors only at right angles or straight ahead; instead of a real amino acid's complex of properties, a mer can be assigned just a few.
"Lattice models aren't meant to model specific proteins," says Rokhsar, "but they give a good representation of certain aspects of real processes in manageable time." Using the Cray T3E at the National Energy Research Scientific Computing Center (nersc), Rokhsar and Vijay Pande, an assistant professor of chemistry at Stanford University, discovered unsuspected regularities in the folding pathways of model polymers.
When the simulated temperature was raised high enough, their lattice model unfolded completely; when the temperature was lowered, the model refolded, writhing through almost a million different positions before settling into its native, low-energy structure. Even with a 48-mer model-roughly equivalent to a small protein-the possible initial conformations are astronomical, and each path to stability is potentially unique.
Rokhsar and Pande analyzed movies made by grabbing snapshots of the writhing polymer every 10,000 iterations. At first the unfolded polymer fluctuates wildly through hundreds of thousands of configurations-then suddenly settles into a partially folded intermediate state, in which a stable core is accompanied by flailing loops and dangling ends. After another couple of hundred thousand iterations, the polymer abruptly locks into its native state.
The model exhibited more than one distinct class of transition state-different substructures that achieve temporary stability at an energy higher than the native state and represent different folding pathways. To see how different properties of the components may affect transition states and pathways, Rokhsar, Pande, and graduate student Nicholas Putnam designed three other small, 27-unit polymers with the same native-state conformation, based on three widely used types of lattice models.
In the simplest version, only mers that touched in the native state attracted each other-all others were energetically neutral. A more complex model had three kinds of mers in competition, with like types attracting one another more strongly than unlike types. The most complicated lattice model used mers with 20 discrete values derived from those of real amino-acid residues.
"In the two simpler cases, we found that folding pathways could pass through just two distinct core transition states," says Rokhsar. "The more complex model had only a single transition state. Both these behaviors are observed in the folding of some small natural protein structures."

|
 |
 |
 |
|||
| Lattice models created by Daniel Rokhsar and Vijay Pande, using the Cray T3E at the National Energy Research Scientific Computing Center (NERSC), have revealed unexpected regularities in the folding pathways of protein-like structures. | ||||||
|
 |
 |
 |
|||
Knowing more about the transitional structures that a folding protein must pass through sheds light on which positions in the chain of amino-acid residues are most critical for a flawless fold-those positions where mutations that substitute one amino acid for another are likely to have the greatest effect on a protein's shape, for better or worse.
Water,
Water, Everywhere
Proteins don't exist as ideal Platonic forms; their real environment consists mostly of a warm solvent, namely water. By combining theoretical and computational approaches, such as lattice models, with data from experiments, physical chemist Teresa Head-Gordon of the Physical Biosciences Division and her colleagues have detailed water's essential role in driving protein folding and stabilization.
"Protein folding in a water environment is hard to model, but it's unavoidable if we're to understand what really happens in nature," says Head-Gordon. "Water itself is an unusual liquid, still far from completely understood. And the behavior of water molecules close to amino acids is markedly different from the behavior of water in bulk."
One important measure of amino acids is their varying degrees of hydrophobicity, or "fear of water." Oil is hydrophobic-that's why oil drops remain separate in water-while hydrophilic ("water-loving") substances readily dissolve in it. Many proteins have a hydrophobic core and a hydrophilic surface.
"Nine of the 20 amino acids that form proteins are hydrophobic," says Head-Gordon. "We started by studying leucines, which are found in the hydrophobic cores of many proteins, and used x-ray scattering experiments to determine what correlations exist among leucine molecules dissolved in water-first at low concentrations, then at higher concentrations."
By measuring the intensities of x-rays or neutrons scattered by water molecules alone-and then by leucine molecules dissolved in water-Head-Gordon and her colleagues were able to analyze the structure of water near the leucine. They conjectured that these water structures, much more highly ordered than water in bulk, give rise to forces that differ among different kinds of amino acids and thus influence folding pathways.
"Hydrophobic amino acids like to be in contact," says Head-Gordon, "but we found that, rather than water being instantly driven out as leucine molecules come together, there is also a preference for the leucines to be separated by a structured layer of water molecules. This forms a gel-like intermediate state which allows forces other than hydrophobic forces to come into play"-other forms of atomic and molecular attraction and repulsion-"as the core takes shape and the protein folds into its native state."
When Head-Gordon and her colleagues applied what they had learned from scattering experiments to lattice models of polymers, they found that by including accurate solvation forces they could go a long way toward making the models more realistic mimics of actual proteins. Some models were swiftly eliminated, and the performance of others was improved to exhibit faster folding and more cooperative folding transitions. In addition to a basic understanding of the folding of all proteins, such studies may lead to specific insight into classic sequences such as the "leucine zipper" that joins secondary protein structures into dimers through hydrophobic attraction-a sequence that, when mutated, may play a prominent role in activating cancer-causing genes.
SCOPing
Out Folds
 |
|
Simple theoretical models bolstered by experimental data are one approach to faster protein-structure prediction. Another way to use computers to translate dna sequences into protein structures is to work directly from a growing library of known folds.
"Structure is of purely scientific interest; function is why people care," says Inna Dubchak, a computer scientist in nersc's Center for Bioinformatics and Computational Genomics. "I use knowledge-based methods to apply what we already know about the properties of molecules to predict the structures of unknown proteins-information that biologists can use to deduce their functions."
Describing her method of predicting the folds of unknown proteins, Dubchak explains that "traditional methods compare unknown gene sequences to known protein sequences or structures residue by residue, searching for correspondences. But what happens when no similar sequence exists? I decided to tackle the problem differently, from a taxonometric perspective."
Dubchak assessed the physical properties of each of the 20 amino acids found in proteins-such characteristics as hydrophobicity, polarity, van der Waals radius (size), and the like-and reduced these to a number of vectors representing the residue's cooperative influence on a fold.
Taken together, the vectors of an unknown sequence do not specify an exact shape so much as they suggest one that may or may not resemble a fold already included in the Structural Classification of Proteins (scop), a library of experimentally observed folds developed by the Medical Research Council's Laboratory of Molecular Biology in Cambridge, England.
Dubchak "trains" neural networks, built with computer processors, to recognize sequences that produce scop-like folds; at present, about a fourth of new sequences can be matched confidently to folds already in the library. Those that don't match known shapes represent folds that have not yet been discovered (or they signal that the neural network doesn't have enough information or hasn't yet learned to recognize the relationship).
Armed with the knowledge that the fold of a new protein resembles familiar folds, biologists can hypothesize the new protein's evolutionary relationships and biological functions, as well as how it may bind to other proteins and to specific chemicals, including drugs.
"We want to focus on the most important projects from the biologists' point of view," Dubchak says. "We want to help biologists solve their hardest problems by applying computational methods."
However, because entirely different dna sequences may produce structures of similar topology, large uncertainties remain. For example, the resolution of a neural-network fold prediction may be limited to several times the typical distance between atoms-and two structures possessing the same fold may be significantly different in size.
 |
|
| Using global optimization programs such as GOSPEL, small protein structures like the alpha chain of uteroglobin can be predicted from sequence (right)-coming close, but not quite matching the crystal structure determined by experiment (left). | |
Teresa Head-Gordon seeks to reduce these uncertainties by invoking the gospel-that is, "global optimization strategies to probe energy landscapes." Head-Gordon's goal is to find, within the range of possibilities, the protein structure corresponding to a specific sequence that has the lowest energy.
Neural-network predictions such as Dubchak's supply "soft constraints" on shape and specify known secondary structures such as alpha helices and beta sheets. By applying gospel -using force-field models such as amber and charmm, and descriptions of aqueous solvation learned from theory and experiment-vaguely defined "coil" structures, which are more challenging, can also be resolved.
In the course of comparing candidates, the algorithm applies these empirically derived functions to areas of the fold accessible to water; it imposes an extra energy penalty on structures with exposed hydrophobic surfaces. Repeated perturbations of amino-acid positions use gospel to lower the energy further, homing in on the lowest possible total energy.
Global optimization is a voracious consumer of computer power and time. Using the Cray T3E-900 at nersc, Head-Gordon and her colleagues have tested their algorithm against simple "target" proteins. In the case of 1pou, for example, a dna binding protein with 72 amino acids arranged as several alpha helices, the structure predicted by gospel from sequence gave a reasonable estimate of the fold but had some six percent higher binding energy than the known structure derived from nuclear magnetic resonance imaging.
"We have still not reached crystal structure energy yet, so further improvements in structure are still possible!" Head-Gordon exclaims.
Nevertheless, while improvements in the underlying model are needed, global-optimization results have been sufficiently encouraging to attempt larger proteins with more complex structures, including pure beta sheets and mixed alpha-helix, beta-sheet proteins.
Bundles
and Beads and Barrels and Saddles
 |
|
 |
|
 |
|
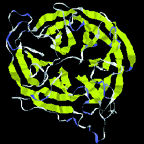
| Protein shapes reveal recurring structural motifs called "folds" that help define physical and chemical properties. Commonly seen folds include beta-sheets or ribbons (top two images), alpha-helices (third from top), and complex globular conformations (bottom). | |
Proteins are like strings of beads wound into bundles. Their structure is described at increasingly intricate levels. w Primary structure is a chain of amino-acid residues, chemical units linked to their neighbors by peptide bonds, like snap-together plastic beads. The 20 amino acids that can form proteins differ in size, shape, electric charge and polarity (which affects interaction with water), hydrophobicity ("oiliness"), and other properties. Researchers have assigned single-letter designations to each, from A for alanine through Y for tyrosine; thus primary structure, the polypeptide chain, is given by a string of letters, e.g., MEIMKKQNSQINEINKDEIFV. . . .
Secondary structure results from the angles between amino acids, plus the hydrogen bonds that may form from one residue to another. Repeating bonds and angles commonly form alpha helices and beta sheets (or sometimes variations of these) and their hairpin or crossover connections-plus a variety of turns, which often expose active chemical groups on the protein surface, and a few other structures such as loops and "paperclips."
Tertiary structures are made from helices, sheets, and other secondary elements. A particular configuration of these is called a fold. There are roughly 500 known folds, a dozen of which occur very commonly, some with names like "barrel" or "sandwich" or "saddle"-out of some 6,000 to 10,000 predicted to exist. Remarkably, many proteins that have completely different sequences of amino acids are structurally identical-a strong hint that this structure has inherent evolutionary advantage. w
While a protein may consist of a single polypeptide strand incorporating
a particular fold, others are built from separate strands. A famous example
of quaternary structure is hemoglobin, which combines two pairs of identically
folded chains in a single molecule capable of snapping up, carrying, and
releasing oxygen in the bloodstream and tissues of the human body.
In vivo, In vitro, In silico
Models that derive values from real amino-acid residues and realistic watery environments can help us understand the folding of real proteins, and the shapes and functions of many unknown proteins can be deduced from libraries of known folds. These and yet more sophisticated and powerful computer techniques are essential, for a functioning protein is dynamic, while the protein structures determined by crystallography are static-and even at the present rapid experimental clip it could take another century to decipher the full atomic structures of all the proteins in cells by experiment alone.
Daniel Rokhsar and his colleagues have also studied the molecular dynamics of a real protein structure, not under natural conditions or in an experimental set-up, but in silico, using a fully realistic "all-atom" computer model in which the properties of every atom in every amino acid are represented, and thousands of water molecules are explicitly treated.
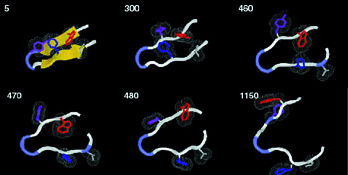
"Even in long runs on powerful computers, with all-atom calculations it's only practical to model a few nanoseconds of real time," says Rokhsar, "yet real proteins typically fold up in a few milliseconds"-a million times longer. "So we modeled a very small part of a real protein, a common structure called a beta hairpin. Instead of trying to watch it fold up, we watch it unfold, which at the high temperatures of the simulation is a much quicker process."
Unfolding occurs in a series of discrete steps which always happen in the same order. Each represents the dissolution of a specific part of the hairpin structure, recalling the transition states of lattice models.
Much faster and more manageable supercomputers will be needed to study larger protein structures at the atomic level. The largest yet studied in silico, with 36 residues and 12,000 atoms, was tracked over the course of a single microsecond by researchers at the University of California at San Francisco; the simulation took a Cray T3D and a Cray T3E-600 running for two months each, and the model did not reach the real protein's native conformation.
 |
|
| At 400 degrees Kelvin, a protein's beta hairpin, 16 amino-acid residues long, starts to unfold. The time to each step of this all-atom simulation is shown in trillionths of a second. | |
To rationally design drugs that can attack specific disease mechanisms, to create novel industrial enzymes, to engineer new organisms that can increase food production, clean up waste, and restore the environment-these potential benefits all depend upon accurate, intimate knowledge of a wide range of protein structures and their possible mutations. Every scrap of experimental knowledge, every advance in calculating the molecular dynamics of model proteins, all are essential to the solution of the protein folding problem, a goal that still glimmers in the future. - end -
| < Research Review | Top ^ | Next > |