
TO TEST POWDERS FOR PROPERTIES OF SCINTILLATION, Derenzo and Moses require a source of x-rays. Searches by their predecessors relied on facilities that produced continuous x-ray beams. While a continuous beam can reveal whether a material scintillates, and its brightness and stopping power, it does not reveal how fast a material is. To sidestep this problem, researchers would actually shoot a bullet across the path of the beam, recounts Moses. This would interrupt the beam long enough to allow a crude measurement of the response time of the material.
In 1988, the LBL researchers moved to a technique that is literally faster than a speeding bullet. They began testing prospective scintillators, first at the Stanford Synchrotron Radiation Laboratory and then at the National Synchrotron Light Source at Brookhaven National Laboratory in New York. Brookhaven's NSLS allowed researchers to put away their guns: Its x-ray beam pulses are only one nanosecond (a billionth of a second) in length.
More recently, the scientists moved the search to their own laboratory at LBL, where they have designed and constructed a unique tabletop x-ray device with the help of graduate student Steven Blankespoor and electrical engineer Matt Ho. The x-ray source delivers 0.1 nanosecond pulses, allowing measurement of even faster scintillators than at Brookhaven.
The instrument was developed in a collaborative project with Hamamatsu Photonics of Japan. It combines a state-of-the-art laser diode and a recently developed light-excited x-ray tube. Essentially, the tube is a single-stage photomultiplier with a photocathode and tungsten anode. Light from the laser liberates photoelectrons at the photocathode, which then strike the anode, producing x-rays.
Moses says the technique is fairly simple. "We use the tabletop x-ray source to expose a sample to a short, intense burst of x-rays. Then," he says, "we see if the sample glows. This is done with a photomultiplier tube which counts photons."
So far, Derenzo and Moses have examined roughly 600 materials. About 10 percent of the candidate compounds tested have been bright enough, but of these, only about 10 percent are fast enough. This leaves some eight substances worthy of further investigation.
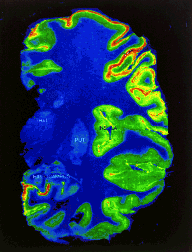
The potential benefits from better scintillators can be dramatic. Take the case of positron emission tomography, important in both diagnostic medicine and research. Among medical imaging techniques, a PET scan is unique in its ability to quantitatively measure biochemical activity as it takes place in the body. PET images show processes like glucose uptake by a cell or blood flow in the brain. They reveal the hidden workings of the human body to researchers while allowing physicians to learn whether a bodily activity is normal or abnormal.
In PET imaging, a positron-emitting radioactive drug is given to the patient. Different drugs concentrate in specific organs or types of tissue. As the radioactivity decays, it emits a positron that encounters an electron and annihilates, creating two gamma rays that travel in 180-degree opposite directions. These pairs are detected by a ring of scintillator crystals that encircles the subject. Computers then draw lines between the two points on the ring where the pairs were detected, lines which pass through the annihilation point in the body. The mathematical technique of computed tomography uses all these lines to produce a computerized image of the area of the body where the tracer has concentrated.

Scintillators play a key role in creating PET images such as the one above showing blood flow in the brain. They reveal the hidden workings of the human body to researchers while allowing physicians to learn whether a bodily activity is normal or abnormal.
Currently, PET images are constructed without an exact knowledge of the time of flight of the photons from their source in the body to the ring of scintillators. The inability to record time of flight is due to the slowness of current scintillators relative to the speed of light. Light travels one foot per nanosecond. If time-of-flight information were incorporated to create a PET image, the fastest current scintillators could pinpoint a location to no better than within 3 inches.
Fast new scintillators must be discovered if time-of-flight information is to be captured to enhance imaging.
Says Derenzo, "If we find subnanosecond scintillators, this will allow us to time the detection of the two annihilation photons so that the physical location of the annihilation point can be pinpointed."
Adds Moses, "Adding the time-of-flight information would be equivalent to washing your windows: you would suddenly have a clean, crisp view."
The LBL researchers have discovered several fast new scintillators that have the potential for use in medicine, science, and industry.
Lead sulfate, which is three times faster than the BGO compound used in PET scans, has properties that make it ideal for a wide range of uses. However, so far, no economical system for growing large, high-quality crystals has been found.
Cerium fluoride and cerium-doped lanthanum fluoride are up to 10 times faster than BGO. While they may ultimately be used in PET scans, these new scintillators currently are attracting the interest of high-energy physicists. One group plans to build an electromagnetic calorimeter with up to 60 cubic meters of cerium fluoride scintillator to study the particles produced by colliding beams at ultrahigh energies.
Derenzo and Moses would like to discover a single compound that combines the best properties of the different scintillators in common usage today. Faster than barium fluoride, brighter than thallium-activated sodium iodide, and with more stopping power than bismuth germanate, the material would become an all-purpose scintillator.
While the researchers don't know whether they will ever find this single ideal scintillator, they expect to find a number of new materials with qualities well suited for specific tasks. Ultimately, they say, their efforts should uncover a host of new scintillators and, along with them, a flash of new discoveries.