 reast cancer is the most common and lethal malignancy in
U.S. women between the ages of 40 and 45. Each year approximately
182,000 American women develop the disease, and each year
approximately 46,000 die from it. What may prove to be an important
discovery in the fight against this disease is a theory postulated by
Berkeley Lab cell biologist Mina Bissell. The theory holds that there
is an important link between the development of breast cancer and a
network of fibrous and globular proteins surrounding breast cells
called the "extracellular matrix" or ECM. Bissell and her
colleagues have shown that the ECM is crucial to the normal
functioning of cells, and loss of or damage to the ECM can lead to
malignancy in transgenic mice.
reast cancer is the most common and lethal malignancy in
U.S. women between the ages of 40 and 45. Each year approximately
182,000 American women develop the disease, and each year
approximately 46,000 die from it. What may prove to be an important
discovery in the fight against this disease is a theory postulated by
Berkeley Lab cell biologist Mina Bissell. The theory holds that there
is an important link between the development of breast cancer and a
network of fibrous and globular proteins surrounding breast cells
called the "extracellular matrix" or ECM. Bissell and her
colleagues have shown that the ECM is crucial to the normal
functioning of cells, and loss of or damage to the ECM can lead to
malignancy in transgenic mice.
Since it was first announced in 1982, the ECM theory has gained
support from experimental results. This past spring, however, it was
put to its most stern test. Bissell and Valerie Weaver, a
post-doctoral scholar in cell and molecular biology, worked with
unique lines of human mammary epithelial cells: one that remains
normal, and one that becomes malignant following a known sequence of
events. The malignant strain was treated in culture with an antibody
that blocks one of the integrins-proteins which reside in the outer
membrane of a cell and through which signals from the ECM are
transmitted inside.
 |
(Click on image to enlarge)
|
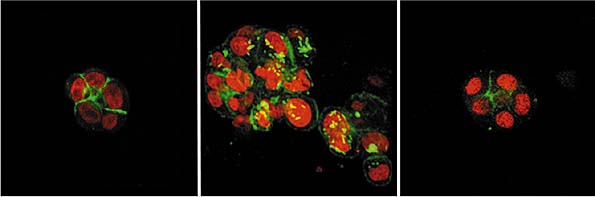
| This
image shows a cluster of normal breast cells on the left, tumor
cells in the center, and reverted tumor cells on the right. The
reverted cells were treated with an integrin-blocking antibody.
Areas shown in red are cell nuclei, the green areas are cyto-skeletal
protein. These images were produced by Carolyn Larabell. |
Under the influence of the integrin-blocking antibody, the malignant
cells reverted back to a normal appearance, their cancerous growth
ceased, and they functioned as if they were healthy cells. Injected
into mice, the same type of treated malignant cells yielded far fewer
tumors that grew much more slowly than untreated cancer cells. By
altering the pathways between the ECM and the nucleus, Bissell and
Weaver were able to revert malignant cells back into almost normal
form and function. When the reverted cells were examined with a
fluorescent probe, the genes in their nuclei were still abnormal,
indicating that the phenotype had been changed but not the genotype.
"These findings have vital implications for breast cancer
diagnosis and prognosis," says Bissell. "Cancer is the
result of not just genetic change, not just developmental regulation,
or loss of growth regulation, but an interweaving of all of these
factors."
The results of this experiment, even if in only one tumor cell
population, help explain why breast cancer takes so long to develop
even in women who are at high genetic risk. So long as cellular and
tissue structure are maintained, tumor development can be suppressed
despite the presence of cancer-inducing genes. Over the course of four
decades in a woman's life, the ebb and flow of hormones and other
contributors to a cell's microenvironment and genome take their toll.
Eventually, through mechanisms not yet identified, the ECM is altered.
This, in turn, alters the signals sent from the ECM into the cell's
nucleus, and, if there are harmful mutations, the sum triggers the
onset of malignancy.
| Lead scientist on this project |
|
|
|
|
|
As a further proof-of-principle, Weaver, Bissell, and their colleagues
also worked with the normal strain of their human breast cells. Again,
altering cell microenvironments with integrin-blocking antibodies,
they demonstrated that normal cells could be induced to look and act
like cancer cells in culture.
Although this research does not offer a cure for breast cancer, it
does point to the possibility that the disease may be treated or
perhaps even prevented through means other than conventional genetic
therapy.
"We've shown that even after cancer genes have been expressed
and lesions have formed, it may still be possible to reverse the tumor
process and restore cells to normal appearance and function,"
says Bissell.
For additional information, also see the Bissell
Lab website.
- Lynn
Yarris



 reast cancer is the most common and lethal malignancy in
U.S. women between the ages of 40 and 45. Each year approximately
182,000 American women develop the disease, and each year
approximately 46,000 die from it. What may prove to be an important
discovery in the fight against this disease is a theory postulated by
Berkeley Lab cell biologist Mina Bissell. The theory holds that there
is an important link between the development of breast cancer and a
network of fibrous and globular proteins surrounding breast cells
called the "extracellular matrix" or ECM. Bissell and her
colleagues have shown that the ECM is crucial to the normal
functioning of cells, and loss of or damage to the ECM can lead to
malignancy in transgenic mice.
reast cancer is the most common and lethal malignancy in
U.S. women between the ages of 40 and 45. Each year approximately
182,000 American women develop the disease, and each year
approximately 46,000 die from it. What may prove to be an important
discovery in the fight against this disease is a theory postulated by
Berkeley Lab cell biologist Mina Bissell. The theory holds that there
is an important link between the development of breast cancer and a
network of fibrous and globular proteins surrounding breast cells
called the "extracellular matrix" or ECM. Bissell and her
colleagues have shown that the ECM is crucial to the normal
functioning of cells, and loss of or damage to the ECM can lead to
malignancy in transgenic mice.

