

That the genetic process can sometimes go awry is nowhere more evident than in the onset of cancer. Among all the terrible forms of this disease, there is perhaps none more fearsome to women in this country than breast cancer. It is estimated that about one in eleven of all U.S. women will develop breast cancer. Approximately one-third of these women, when treated with current therapy, will survive for about 20 years after diagnosis.
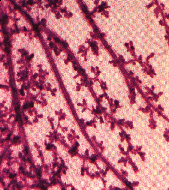 Carmine staining of mammary glands in transgenic mice
showed that breast cancer can occur from the breakdown of the "extracellular
cellular matrix" a mass of proteins that give strength and texture to cells.
Carmine staining of mammary glands in transgenic mice
showed that breast cancer can occur from the breakdown of the "extracellular
cellular matrix" a mass of proteins that give strength and texture to cells.
At LBL, researchers have been studying the processes by which cells carrying
identical genetic programs are able to differentiate at some point during their
development to become a specific type of tissue or organ. They have then been
applying their findings to understanding how this specificity is lost during
malignancy. A major discovery was the demonstration that the microenvironment
of the cell, including a mass of fibrous and globular proteins called the
"extracellular cellular matrix" (ECM) which surrounds a breast cell, plays a
vital role in cell growth and development and, consequently, cancer. The
biology and biochemistry of a normal cell and a cancerous cell have many common
features. The difference is that cell division in the cancerous cell runs
rampant. In addition, the cancer cell apparently is unable to sense the
microenvironmental cues. Starting with the ECM discovery, LBL scientists have
build up a picture of the interaction between breast cells and ECM, which was
presented this past year at a special symposium sponsored by the American
Association for the Advancement of Science. A highlight of the presentation
was the announcement of the first three-dimensional rapid assay for
distinguishing normal from malignant human breast cells in tissue culture.
This simple but informative assay should serve as a powerful tool in breast
cancer research. Already it has been used at LBL to provide evidence that a
"tumor suppressor gene" indeed is capable of arresting the growth of metastatic
tumor cells in the 3-D assay.
After the question of why a normal cell turns cancerous, the other big mystery in cell biology is why does a normal cell turn senescent--meaning it remains metabolically active but no longer divides. Scientists know senescence is an irreversible stage in the life of the cell but want to learn more about it because--as the reverse of cancer--its secrets might help prevent the uncontrolled growth of a malignancy. This past year, LBL scientists were instrumental in developing the first simple, rapid, and inexpensive test for identifying senescent cells that can be used on living organisms. Until now, senescence research has been limited to the study of cells grown in culture. The new test was developed after researchers discovered that senescent cells produce an enzyme called beta-galactosidase that is virtually absent in normal cells. Based on this finding, a simply assay was developed that uses a stain to detect the presence of the enzyme. Old cells--those that express beta-galactosidase--turn blue. The ability to identify senescent cells on living creatures will allow researchers to take a much closer look at the role of these cells in tumor suppression. Now scientists can screen compounds for senescence-inducing or senescence-delaying activities that could have anti-tumor or anti-aging properties. The test can also be used to identify genes that might be able to stop cancerous cells from replicating, and genes that trigger premature aging.
Of course many deadly diseases are not the result of a breakdown in the genetic process, but are introduced into our bodies by external elements. One of the oldest and most persistent of these diseases is malaria which was described by Hippocrates in 5 B.C. According to the World Health Organization, each year between 300 to 500 million people living in tropical or subtropical regions of the world will become infected and suffer the burning fever and severe pain that the disease inflicts. Nearly three million malaria victims will die.
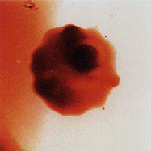
An intact red blood cell infected by the malaria parasite, imaged by the x-ray microscope at the Advanced Light Source. The microscope is dedicated to high resolution microscopy for biology and materials sciences.
Malaria is caused by infection of red blood cells with a parasite called
"plasmodium" which is transmitted through the bite of a female anopheles
mosquito. Drug therapies and insecticides have been brought to bear against
this ancient scourge, but resistant strains of both the parasite and the
mosquito continue to emerge. At LBL, researchers have been studying the
deadliest strain of the parasite, seeking to better understand its life cycle
in the human blood stream and, in particular, exactly how it defeats the body's
natural defenses. With this knowledge, medical researchers will be better
equipped both to prevent and to heal infections. This past year saw the
start-up of an x-ray imaging microscope at LBL's Advanced Light Source. Working
with laser-like beams of x-rays that can be used to look deep inside living
cells, the researchers were able to produce one of the clearest, most detailed
images ever obtained of a red cell infected with the malaria parasite. With the
accumulation of more of these images, the researchers expect to acquire a
complete picture of the pathology behind malaria.
All living cells on this planet start with the genetic code that is stored within the spiraling coils of the DNA molecule. The ability to decipher this code has been called the "Holy Grail of Biology" for what it would mean to biological and medical research. Hence the establishment of the Human Genome Project by the U.S. Department of Energy and the National Institutes of Health. To appreciate the enormity of this task, however, consider that if all of the DNA in the nucleus of a typical human cell were to be unraveled and stretched out flat, it would extend about 180 centimeters (six feet) in length. After seven years of national effort, scarcely a centimeter (half-an-inch) of this DNA has been deciphered. Everyone involved with the Human Genome project agrees that the process must be speeded up for the project to be completed within a reasonable period of time and at an affordable cost.
At LBL, which is home to the one of three centers for human genome research established by DOE, a key segment of the program is focused on finding ways that will bring the project to a swifter completion. This past year, researchers announced positive results with what they call a "directed approach" to determining the sequence of the base-pairs that make up the letters in the genetic code. The technique involves the creation of sequencing templates that have been "mapped" (located on a chromosome) to a resolution of 30 base pairs prior to being sequenced. This high resolution mapping information provides two major advantages over previous sequencing techniques: sequences can be determined with far fewer steps; and the difficulty of putting sequences together is greatly reduced. Working with their directed approach, two teams totaling 12 members were able to sequence 2 million base-pairs by the end of 1994. Directed-sequencing is expected to fit in well with the efforts of other LBL researchers to automate the genome project. All of this work should benefit from the inclusion in President Clinton's 1994 budget proposal of $2.2 million to begin construction on a Human Genome Laboratory at LBL.

Return to the Table of Contents of the 1994 Regents Report
