| "Industrial carbon sequestration is an
extreme option for mitigating greenhouse gases," says Sally Benson,
director of Berkeley Lab's Earth Sciences Division (ESD), who notes that
the Department of Energy's traditional approach to carbon management has
emphasized energy efficiency, low-carbon fuels, and alternative energy
sources.
Three years ago, however, when the President's Committee of Advisors on
Science and Technology urged DOE to pay more attention to carbon
sequestration -- to taking carbon emissions from power plants and other
sources out of circulation -- Benson became one of the leaders of the new
research emphasis. "I see this as an opportunity to prepare for the
time when people realize that deep reductions in greenhouse gases are
needed."
In 2000, ESD became host to two major DOE sequestration programs. Jim
Bishop, with Ken Caldeira of Lawrence Livermore National Laboratory,
codirects the DOE's Center for Research on Ocean Carbon Sequestration.
Benson directs the GEO-SEQ Project supported by the Office of Fossil
Energy.
GEO-SEQ (pronounced "geo-seek"), which is managed by ESD's
Larry Myer, is a partnership of government agencies, academic
institutions, and private energy companies in the U.S. and Canada
investigating sequestration in geological formations. "Geological
sequestration involves taking CO2 that has been separated from industrial
flue gases and pumping it into underground formations," Benson
explains.
"C02 currently makes up five to 15 percent of power plant
emissions, depending on the type of power plant. Unfortunately, to
separate it using existing processes costs $20 to $70 a metric ton,
depending on the facility." Therefore an important research goal is
to develop better and cheaper separation and transportation techniques.
However, says Benson, "the economics change if we can use
sequestered CO2 to recover more fossil fuels." Carbon dioxide can
help extract oil and gas from depleted reservoirs and can increase the
production of methane, the chief component of natural gas, from coal beds
that can't be mined.
CO2 helps recover more oil
 |
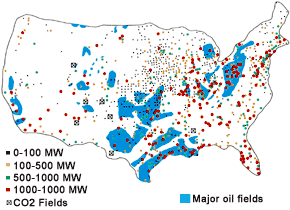
LOCATIONS OF MAJOR OIL FIELDS, POWER
PLANTS, AND CO2 FIELDS
|
The petroleum industry has long used CO2 injection to get more oil from
depleted reservoirs. If the pressure is high enough in these formations,
the CO2 and oil become completely miscible, leading to highly efficient
oil recovery.
At lower pressures CO2 displaces oil without mixing together to form a
single fluid phase -- this is known as immiscible displacement. This too
enhances recovery, by reducing the oil's viscosity and by swelling, as
some fraction of the CO2 dissolves in the oil. While some of the carbon
dioxide comes back up with the oil, much remains underground. Operations
can be modified so that more of the CO2 remains underground after the
enhanced-recovery project is complete.
"One of our partners in GEO-SEQ is Chevron, which is doing a pilot
project at the Lost Hills Oil Field in California�s Central Valley. They
aim to enhance oil recovery in the Diatomite Formation by injecting
CO2," Benson explains. Diatomite, which consists of the fossilized
skeletal remains of aquatic plants, has been called a "glass
sponge" -- it holds a lot of oil, but the oil is very difficult to
get out.
 |

CHEVRON LOST HILLS TEST SITE WITH CO2 INJECTION WELL SHOWN
TO LEFT OF WORKER IN FOREGROUND
|
"Immiscible displacement of oil by CO2 injection is one of the
ways they are trying to recover more of the oil in this formation,"
she says. "This provides a great opportunity for us to develop and
demonstrate geophysical imaging methods." ESD�s Ernie Majer, Tom
Daley, and Mike Hoversten have completed the first round of data
collection at Lost Hills and are awaiting post-injection surveys; by
comparing pre- and post-injection images, they will be able to evaluate
how well their high-resolution seismic and electromagnetic techniques can
track migration of CO2 deep underground.
More energy from gas and coal
Natural gas fields have shown that they can store gases for millions of
years; thus they are promising targets for CO2 sequestration. "As an
added benefit -- although we haven't tested the idea yet -- we are working
on the concept of prolonging production in depleted gas fields through
carbon dioxide injection," says Benson.
Recently she worked with ESD's Curtis Oldenburg and Karsten Pruess to
build a computer model of the depleted Rio Vista gas field in the
Sacramento River Delta, the largest onshore field in California. Their
study indicated that by injecting the field with carbon dioxide from a
nearby gas-fired powerplant in Antioch, it might be possible to return
methane production, now minimal, to 40 to 80 percent of peak historic
levels for at least 5 years, and perhaps for 10 years or longer.
The researchers used the TOUGH2 reservoir-simulator program developed
by Pruess and his ESD colleagues. Benson notes that "our long
experience modeling underground flows of all kinds is one of the things
that makes Berkeley Lab particularly well suited to the investigation of
geologic sequestration."
Unminable coal beds are another likely place to store carbon. Methane
adsorbed in coal is familiar as deadly mine gas, a cause of numerous
tragedies; formerly discarded, it now accounts for some five percent of
the country's natural gas production. A pilot project in San Juan, New
Mexico suggests that methane production from the extensive coal beds there
could be increased 75 percent by injecting carbon dioxide, which frees and
replaces trapped methane.
The salt of the earth
"It is estimated that there is enough capacity in oil and gas
reservoirs to sequester many decades of the world's CO2 emissions from
power plants and other industries," Benson says -- but with much
greater capacity, "deep brine-filled formations could store the
world's carbon emissions for centuries."
Storing carbon in brine formations gives back no fossil fuel, but there
are economic incentives nevertheless. For example, natural gas from the
Statoil company's Sleipner West field in the North Sea contains too much
carbon dioxide for commercial use, and Norway taxes CO2 emissions from
off-shore production. So each year a million metric tons of carbon dioxide
are separated from the gas and injected into a brine formation deep
beneath the seabed. In the United States, brine-filled formations may be
able to sequester a half a billion metric tons of carbon dioxide.
Cutting costs
"Identifying the sequestration capacities of geological formations
is just one of our goals in GEO-SEQ," says Benson. "We're also
studying ways to lower the overall cost of geologic sequestration by
looking for opportunities to reduce the cost of transportation and CO2
separation. We want to develop and apply advanced reactive-transport
models, to identify sequestration sites near our major power plants, and
to find the optimal degree of CO2 separation. If we learn that some amount
of trace gases can enhance the sequestration process, or at least not
interfere with it, the cost of separation may drop dramatically."
And, she says, "we need monitoring technologies to assure us that
once the CO2 is stored, it's stored safely and permanently." In the
long run, this may be the most important goal of all, the one that
determines whether the public accepts the concept of carbon sequestration.
"With ocean and terrestrial sequestration, we need to understand
ecosystem impacts and long term effectiveness. With geological
sequestration, we need to know that the carbon we put in the ground isn't
going to come back up," says Benson. "Otherwise we're just
transferring our responsibility to future generations."
Additional information:
|