| A new technique based on a bright,
tightly focused beam of infrared light from the Advanced Light Source (ALS)
at the Department of Energy's Lawrence Berkeley National Laboratory allows
researchers to follow subtle chemical and molecular changes in individual
human cells, without killing the cells or using intrusive probes.
"Traditional methods of biomedical research either require killing
cells" -- known as fixing them -- "or averaging results from
many cells, or introducing dyes or tagged proteins or other agents that
can affect cell chemistry -- methods that usually involve tedious sample
preparation and long delays between experiment and result," says
Hoi-Ying Holman of Berkeley Lab's Earth Sciences Division, the principal
investigator in developing the new technique. "Now we can study
individual cells in real time without introducing extraneous
factors."
Holman and her colleague Michael C. Martin discussed their work in
separate talks at the annual meeting of the American Chemical Society in
San Francisco and the American Physical Society in Minneapolis. They
describe how, using SR-FTIR spectromicroscopy -- Synchrotron
Radiation-Based Fourier Transform Infrared spectromicroscopy -- a
technique previously used for studies in environmental, forensic, and
materials sciences, they and their collaborators were able to characterize
changes in living human cells.
Despite the jawbreaking name, the principle is straightforward.
Different molecular and physical states of a cell absorb different
wavelengths of infrared light; light transmitted by the cell yields a
unique spectrum that can distinguish different cell types, different
phases in the cell cycle, and different chemical reactions and physical
changes inside the cell. One result is that the unique method can identify
and monitor the progress of diseases in human cells.
Key to success is the quality of the synchrotron light from beamline
1.4.3 at the ALS: with hundreds of times the intensity of conventional
infrared sources, the beam can be focused to a spot less than 10
micrometers in diameter (10 millionths of a meter), a little smaller than
the dimensions of a typical mammalian cell.
"We can position the spot on a sample within one micrometer,"
says Wayne McKinney of the ALS, who developed the infrared beam line with
Martin. "Because the synchrotron light comes in pulses two
nanoseconds apart, we can record very fast changes in cells."
A nanosecond is a mere billionth of a second, and although this
capacity has not been applied to work done so far, in the future it
promises to open new insights into cellular processes.
In recent work, Holman and her collaborators have concentrated on the
response of cultured human cells, including lines originated from lung and
liver tissue, to low doses of environmental agents.
"We studied changes in cells caused by oxidizing agents in dilute
amounts typical of environmental exposure," Holman says.
"Hydrogen peroxide is a strong oxidizer, and bleomycin is an
antibiotic that is a weaker oxidizer but still damages DNA."
Hydrogen peroxide causes predominantly single-strand breaks in DNA,
while bleomycin induces a large number of double-strand breaks. Under SR-FTIR
spectroscopy, damage from each chemical showed up as distinctly different
spectral changes.
In addition, the cell-wide damage caused by x rays in lung cells
produced a very different spectroscopic signature compared to unexposed
cells.
"We also used the new technique to detect changes caused by dioxin
in liver tumor cells," Holman says. The dioxin molecule binds to a
specific receptor, which then binds to a site on the cell's DNA,
regulating a gene that expresses one of the cytochromes. Cytochromes are
proteins that catalyze the breakdown of aromatic carcinogens and other
organic molecules.
"Increasing the dose of dioxin caused marked changes in the SR-FTIR
spectrum, but increasing a control compound that doesn't bind to that
receptor didn't show these spectral changes," Holman says, indicating
that dioxin's biological influence is related to its interaction with the
binding site; the degree of binding to cellular receptors results in
distinctly dose-dependent changes in spectral characteristics.
 |
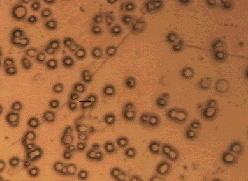
SYNCHROTRON RADIATION-BASED FOURIER TRANSFORM INFRARED
SPECTROMICROSCOPY HAS BEEN USED TO STUDY X-RAY DAMAGE IN LIVING
HUMAN LUNG CELLS LIKE THESE.
|
SR-FTIR spectroscopy also revealed distinctive spectra from individual
human lung cells as they went through different stages of the cell cycle.
Spectra varied during the period preceding DNA synthesis (G1 phase), the
period of DNA duplication (S phase), and cell division (M phase). Among
other factors, the different spectra indicated changes in the physical
packing of long, coiling DNA molecules.
SR-FTIR spectroscopy has not only characterized specific changes in
response to different agents but has distinguished the different responses
of individual cells within a population -- and, in some cases, the
restoration of the original spectra as cells repair damages.
"Other researchers are working with us to study a variety of
effects in living cells," Holman says. Planned research includes the
effects of radiation and drug therapies for malignant brain tumors and the
damage from oxidative stress in such diseases as atherosclerosis,
diabetes, and rheumatoid arthritis.
Only recently applied to the study of living human cells, Synchrotron
Radiation-Based Fourier Transform Infrared spectromicroscopy complements
information obtained from standard cellular assays and, even more
important, allows studies impossible or impractical to accomplish by any
other method.
By mapping biological and chemical reactions as they occur in
individual living cells over a period of hours or days -- in response to
dilute, environmentally relevant concentrations of chemical substances and
radiation -- the new technique enables researchers to perform basic
studies of the life, death, damage, and self-repair of tissues and cells
at the subcellular level.
Additional information:
|