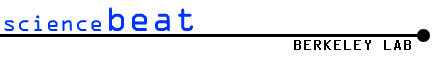|
A team led by Alexander Pines and David Wemmer, working with colleagues
at the Scripps Research Institute in La Jolla, have developed a sensitive
and versatile new biological sensor based on nuclear magnetic resonance
(NMR). Pines and Wemmer are members of Berkeley Lab's Materials Sciences
Division and Physical Biosciences Division respectively; both are professors
of chemistry at the University of California at Berkeley.
The secret of the new biosensor lies in confining laser-polarized xenon
atoms inside specially modified molecular cages. By using optically pumped
xenon, the caged-xenon sensor promises to produce much "brighter"
signals from chemical targets in living organisms. Most current biosensors
use fluorescence, but only a few colors can be used in parallel before
the spectra from different biological molecules overlap and obscure one
another. The caged-xenon NMR sensor, however, can be "multiplexed"
to a high degree, to detect many distinct analytical targets simultaneously.
Several components work together in building different versions of the
versatile biosensor. In all versions, a laser-polarized xenon atom is
trapped in a hollow molecule called a cryptophane. A linker, containing
a short chain of amino acids that makes the cage water soluble, connects
it to a ligand, which can be selected to recognize a specific target of
interest. In another scheme, the linker can connect to a protein whose
binding sites match specific biomolecules to be detected.
|
|
|
|
|
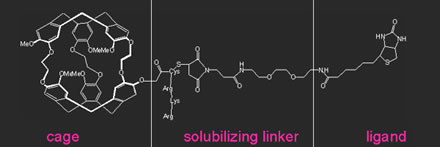
| The xenon biosensor's three main components:
a laser-polarized xenon atom trapped in a cryptophane cage; a linker
with a chain of amino acids to make the assembly soluble in water;
and a ligand to target specific biomolecules |
|
Assembling the NMR biosensor team
"We couldn't have carried out this experiment without the expertise
of different groups," says Megan Spence, whose doctoral dissertation
while a graduate student in the Pines lab was based on the xenon biosensor
research. The goal of the multidisciplinary project was to harness the
remarkable power of laser-polarized xenon in a new way.
To design and craft the "supramolecular cage" assembly, NMR
experts from the Pines laboratory joined with molecular biologists in
Wemmer's group. Expert knowledge of synthetic chemistry came from the
laboratory of Peter Schultz, formerly of Berkeley Lab and UC Berkeley,
now director of the Genomics Institute of the Novartis Research Foundation
and professor of chemistry at the Scripps Research Institute in La Jolla,
Calif.
NMR detects atoms like xenon whose nuclei have a magnetic moment because
there is a slight excess of spin "up" nuclei over spin "down"
nuclei. The excess of up versus down spins is normally only about one
in 100,000, but optically-pumped xenon vastly increases signal strength
by increasing the proportion of spin-up nuclei, producing a population
of xenon atoms with one in five -- some 20 percent or more -- of their
nuclei in the up state.
NMR fingerprints atoms of specific chemical species by detecting their
characteristic emission frequencies after they have been perturbed by
radio waves. Signals from a given type of nucleus vary according to its
chemical environment; bonding in different chemical compounds, for example,
causes distinctive "chemical shifts" in the signal from a single
kind of atom, which show up as separate peaks in an NMR spectrum.
Building the biosensor
The new biosensor is comprised of an arsenal of subtly distinct molecules
with different kinds of cryptophane cages and different kinds of ligands
or proteins, plus different orientations of attached amino acids and tethers.
Because each has a different chemical shift in the NMR spectrum, different
binding events can be simultaneously identified, and are made easily detectable
because of polarized xenon's "brightness."
|
|
 |
 |
|
|
 |
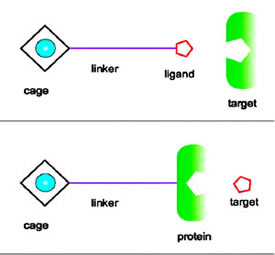
Caged xenon can be linked to a ligand
that binds to proteins; alternately it can be linked to a protein
that binds to specific molecules. |
|
|
However, "xenon is a noble gas and doesn't react, so it takes ingenuity
to use it," Wemmer remarks, noting that the Pines lab has pioneered
laser-polarized xenon NMR in a host of applications, from materials sciences
to medical magnetic resonance imaging (MRI). Wemmer's own group studies
protein structure and function with NMR.
Some proteins have binding sites that can accept xenon in certain conformations;
Wemmer's graduate student Seth Rubin, together with Spence, postdoctoral
fellow Ivan Dimitrov, and graduate student Janette Ruiz of the Pines lab,
had studied weak interactions of free xenon with proteins in solution.
"In some cases, the chemical shifts were larger than expected,"
says Wemmer, which raised the obvious possibility that xenon NMR could
be used to study protein's interactions. One obstacle: "Most proteins
don't have a xenon pocket."
To meet this challenge, Pines, Wemmer, and their coworkers came up with
the idea of using xenon in cages to bind to molecular targets. Wemmer
calls cryptophanes -- invented in the 1980s and not easy to synthesize
"but in the literature" -- an "artificial pocket"
for the xenon. But how to get the artificial pocket to attach to a target
like a protein?
"A classic case of protein binding is the biotin-avidin reaction,"
Wemmer says. Avidin is a protein found in egg whites that binds strongly
to biotin, a B vitamin. "The avidin molecule is like a big magnet
that grabs the smaller biotin." Biotin ligands with chemical tethers
attached are sold commercially; in theory, all that had to be done was
attach the caged xenon to the other end of the tether.
With the major components of the system in place, at least on paper,
and calculations indicating that the sensitivity of the new biosensor
should be excellent, it remained to build the system as designed -- not
an easy task.
The biotin-avidin reaction seemed an excellent testbed for the new sensor.
Convinced of its potential, Peter Schultz and his coworkers, postdoctoral
fellows Shao Qin Yao and Feng Tian, contributed critical techniques and
suggestions, including the incorporation of a short peptide chain of four
amino acids to render the insoluble cryptophane cage soluble in water.
Showing results
To demonstrate the technique, the modified cryptophanes were dissolved
in water, and laser-polarized xenon was added. The cryptophanes quickly
bound xenon atoms, filling essentially all the cages.
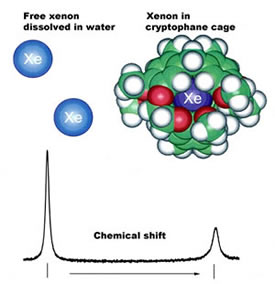
NMR spectra were first taken with no avidin present; the spectra showed
a peak signalling xenon's distinctive chemical environment inside the
modified, water-soluble cryptophane cage -- a peak easily distinguished
from the distant peak of free xenon dissolved in water.
|
|
|
 |
|
|
| The chemical signature of caged xenon
in an NMR spectrum is shifted from that of free xenon. |
|
|
|
When avidin from egg whites was added to the mix, another distinctive
peak appeared in the spectrum. To check that this was a signal of biotin-avidin
binding, a spectrum was taken of a mixture that contained avidin already
saturated with biotin. Under these conditions the peak wasn't present
-- indicating that when present it unambiguously identified caged xenon
bound by biotin ligands to avidin molecules.
"Compared to other good biosensors like fluorescence technologies,
xenon NMR biosensors have two great advantages," says Spence. "With
fluorescence, you have to excite the probes with a laser, so you can't
use them in complex living organisms. But NMR, like its relative MRI,
isn't invasive -- the radio waves used travel easily through cells and
tissue, so analysis can be done in vivo. And the xenon NMR biosensor can
also be multiplexed."
The latter possibility arises not only by choosing ligands which can
bind to different proteins -- or attaching proteins to the xenon cage
which seek out small target molecules instead of other proteins -- but
also because the cryptophane cages themselves can take different shapes
or orientations (chirality), each with a distinct chemical shift.
"This means highly resolved, separate peaks, much easier to read
than comparable spectra from fluorescence, which tend to overlap,"
Spence says.
|
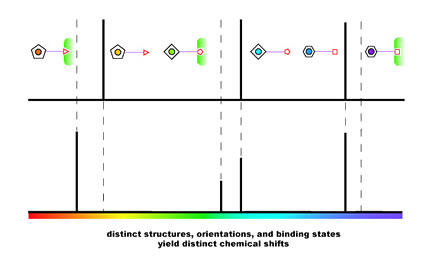
|
|
| A multitude of xenon biosensors with
distinct targets can be distinguished by their chemical shifts in
NMR spectra. |
|
Still other distinguishable molecular structures result from the orientation
of the amino-acid chains and even the orientation of individual atoms
in the tether. "We are only changing directions of atoms, not the
atoms themselves, yet we can build a whole library of chemical shifts
indicating subtle changes," says Spence, comparing this wide variety
of possible simultaneous indicators to the limited range of colors fluorescent
biosensors can use at one time to identify different targets of interest.
Wemmer speculates on the numerous possible uses of xenon biosensors:
"There are lots of times you want to know whether or not something
is present in the body, like arterial plaques or cancer cells, and often
you want to know to what degree something is present compared to something
else. The body can generate antibodies specific to virtually any target.
Maybe we can take that antibody and tie it to xenon in a cage. For any
chemical species for which you can get a selective reaction, you may be
able to use this method."
The technique is being tested with various "diastereomers"
-- several orientations of cryptophanes combined with attached components
of different orientation -- and a variety of ligands and proteins. Development
is still underway and the path to further progress lies open.
"The interesting part is that this research is a hybrid that brought
together a number of different techniques," says Wemmer. "Technique
A was known, and technique B was under development, but when you put A
and B together, you get something never done before."
Additional information:
|