|
 |
 |
|
|
 |
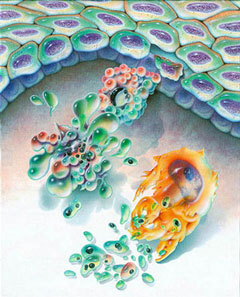
Apoptosis is the orderly process of
programmed cell death, by which organisms remodel their tissues. After
the nucleus and the cell break down, debris is ingested by white blood
cells. Image: NASA |
|
|
Most cells in higher organisms know when it's time to die, for the good
of the whole multicellular being. But tumor cells infamously resist death,
whether from chemotherapeutic drugs or the body's own immune system; finding
out how is a major goal of medical and biological research.
A new study in the journal Cancer Cell (September, 2002) reports
that the formation of cellular structures like those in the breast confers
resistance to cell death in both normal and tumor cells. The study was
conducted by Valerie Weaver of the University of Pennsylvania's Department
of Pathology and Laboratory Science, a member of that university's Institute
for Medicine and Engineering, in collaboration with Mina Bissell of Berkeley
Lab's Life Sciences Division and others; the work had its beginning when
Weaver was a postdoctoral fellow in Bissell's laboratory.
When cells refuse to die
Programmed cell death, or apoptosis, is the process by which organisms
remodel their tissues, ridding themselves of cells infected by viruses,
cells with damaged DNA, and no-longer-needed cells that could become dangerous
-- like immune-system killer cells when the threat of infection is past.
While much is now known about the regulation of apoptosis by specific
genes and proteins within individual cells, tumors remain puzzling. What
allows some tumors to evade cell death while others succumb to chemotherapy
or other treatment?
"Multidrug resistance of tumors is a major problem facing the treatment
of cancer," Weaver says, "but the mechanisms of resistance are
not clear. Cells can be made resistant in two-dimensional cultures --
if sometimes only to one class of drugs -- but when they're put back into
a living body, they lose resistance again."
Weaver, Bissell, and their colleagues found that an important part of
the puzzle involves the way cell structures are organized in the tissues.
Since three-dimensional architectures are lost in two-dimensional cell
cultures, the vital role shape plays in cellular function has long been
obscure.
"Of course researchers understand that tumors grow in 3-D,"
Weaver says. "But critics of studies of multidrug resistance in vivo
say, 'there are so many other things going on in the body, it's not easy
to demonstrate the role of structure.'"
Bissell emphasizes that "cells need to be studied in the context
of their environment, in which growth factors, hormones, and the extracellular
matrix, or ECM, all play vital parts. When forming tissues, you need the
right kind of cross-talk among all these signalling molecules."
A model of living tissue
To study resistance to apoptosis, Weaver, Bissell, and their colleagues
used an exceptional 3-D model of tissue from cultured human breast cells.
The model, initially developed in Bissell's lab for rodents and later
extended to human cells in collaboration with Denmark's Ole Petersen,
now of the University of Liverpool, is capable of distinguishing between
normal and malignant cells. In the 3-D cultured-cell model, unlike experiments
in living organisms, variables can be controlled and studied one by one.
|
|
 |
|
|
| Fluorescent images of key proteins
in 3-D cell structures grown in reconstituted basement membrane: when
their polarity is perturbed, nonmalignant cells lose their resistance
to apoptosis. Disorganized malignant cells, unresistant to begin with,
acquire resistance after being made to "revert" to polar
organization. |
|
| |
|
In the model, nonmalignant mammary epithelial cells -- epithelial cells
are those that form internal and external linings -- form attachments
to a form of extracellular matrix called "reconstituted basement
membrane." In living organisms the basement membrane, consisting
of layers of specialized proteins, is essential to anchor cells in place
in the breast and many other organs.
"The ECM consists of a mass of huge proteins that are secreted outside
the cell," Bissell says. "As far as normal cells are concerned,
it used to be thought that it was needed just to provide a scaffold. But
it does much more: it communicates powerful signals that affect cell behavior
and tissue organization."
The 3-D model includes a reconstituted basement membrane, similar to
that in a living organism, which induces nonmalignant cells to form hollow
spheroids -- "polarized" structures that, as Bissell puts it,
"know which way is up."
Remarkably, the cells in these spheroids are resistant to apoptosis.
By contrast, malignant cells proliferate to form disorganized, nonpolarized
aggregates, susceptible to cell death induced by a variety of drugs and
other agents used to kill cells.
Polar structures of normal cells in the model resemble organoids called
acini (Latin for berries) that secrete milk in mammary tissue. Weaver
emphasizes that it is not necessary to create working acini in order to
confer resistance to cell death: the key is polarity itself.
In the model, formerly disorganized cell aggregates can be made to "revert,"
forming polarized structures through interaction with the reconstituted
basement membrane. Once they have formed spheroids, the malignant cells
also become resistant to apoptosis.
In monolayer cultures, however, both nonmalignant and malignant cells
were equally vulnerable to cell death induced by chemotherapeutic drugs
and immune regulators.
The researchers sought other reasons for the stability of cells in polar
structures, like their growth status -- how fast the cells were dividing.
Unlike what has been believed from clinical studies, however, growth status
did not affect resistance to apoptosis. And since the cultured cells were
genetically identical, whether in monolayer or 3-D, the results indicated
that context can override genetic makeup.
How could structure make such a crucial difference? Polar structures
do not arise in a vacuum. The specific molecular and biochemical pathways
by which cells communicate with one another and attach to the basement
membrane not only influence polarity but hold the key to resistance to
apoptosis.
Cell death, part 2
|