|
|
 |
How does a tiny sulfide particle travel from a Chinese factory to California?
And how does it react when it gets there? Scientists don't know precisely,
which is one of many reasons Berkeley Lab researchers are helping to shape
the future of a new field called nanogeoscience.
As the name implies, it's the study of geological processes involving
particles no larger than 100 nanometers, meaning in some cases as small
as a few atoms across. Such particles play critical roles in carbon sequestration,
air pollution, and even the removal of toxins from soil.
But how, and to what extent, remains a mystery. That's because nanoscale
particles often don't behave like larger solids. A particle's physical
and chemical properties change as it becomes smaller, forcing researchers
to use highly sensitive instrumentation such as transmission electron
microscopes and synchrotron-based spectroscopy to determine how molecule-sized
particles contribute to larger scale phenomena.
And while a small but growing number of scientists probe these poorly
understood processes, their work remains largely isolated. Nanogeoscience
is a new field, and a researcher who studies how nanoparticles aggregate
in soils may not know that other researchers study the analogous process
in oceans.
"People who study aerosols don't talk with people who study oceanic
particles who don't talk with people who study soil particles," says
Glenn Waychunas, a scientist in Berkeley Lab's Earth Sciences Division.
"We need to bring them together."
To shepherd the growth of nanogeoscience research, Waychunas and colleagues
Jillian Banfield, Hoi-Ying Holman, and Paul Alivisatos are planning a
$3 million nanogeoscience program that will encompass Berkeley Lab and
UC Berkeley. The program will complement the lab's Molecular Foundry,
a national user facility opening in 2006 that will allow researchers to
build and test nanoscale devices. And to foster improved communication
between nanogeoscientists everywhere, Waychunas envisions a virtual center
that electronically links several institutions that conduct innovative
research.
|
 |
 |
|
Among these inquiries is learning how oceans capture atmospheric carbon,
a complex process that plays a key role in our understanding of climate
change. Seawater teems with tiny particles that contain nanoscale iron
molecules. And iron is crucial in regulating phytoplankton growth, which,
in turn, influences the exchange of carbon dioxide between the ocean and
atmosphere. So understanding how phytoplankton acquire and use iron will
also help explain how oceans capture atmospheric carbon. But researchers
don't know where these recently observed nanoscale iron molecules originate,
their importance, and how or if organisms use them.
"There is a black hole," says Waychunas. "Oceanographers
generally study particles that measure 0.2 microns and larger, which means
a lot of nanoscale particles are not examined, particularly with respect
to formation mechanisms."
On land, researchers study how nanosized minerals capture toxins such
as arsenic, copper, and lead from the soil. Facilitating this process,
called soil remediation, is a tricky business. Some toxins weakly adhere
to particles through electrostatic forces while others precipitate onto
a particle's surface, forming chemical bonds that are more difficult to
undo. Even more troublesome is aggregation, in which the precipitated
toxin is sandwiched between the grains of a particle. As Waychunas explains,
learning how these processes work on the nanoscale could greatly improve
the effectiveness of remediation efforts.
Another promising soil remediation strategy links nanogeoscience with
biomimetics, an equally new field in which researchers design substances
that mimic biological processes. For example, researchers are interested
in natural processes in which microbes produce minerals that are highly
reactive and therefore ideal components of a toxic uptake system. Taking
this a step further, they hope to manipulate microbes into producing nanosized
minerals of specific sizes and shapes that most efficiently bind with
toxins.
Other research explores the changing characteristics of particles as
they become smaller. At the nanoscale, for example, there are many more
atoms associated with the surface of a particle than the interior. This
intrigues researchers like Waychunas, because most chemical reactions
occur on the surface of minerals, large and small. But what's true for
a fist-sized chunk of quartz doesn't necessarily translate to a particle
only a few molecules across. At this minute size, surface properties can
change, meaning the nature of these all-important surface reactions also
changes.
|
 |
 |
|
|
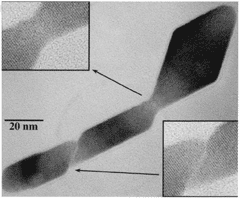
| A transmission electron microscope
image of an anatase (titanium oxide, TiO2) mineral crystal hydrothermally
coarsened in dilute hydrochloric acid. The crystal was assembled from
smaller nanocrystals via a process called oriented attachment, an
alternative to classical growth that occurs on the nanoscale. |
|
|
The list continues. Recent research suggests that some airborne nanoparticles
are so small they zing through the upper atmosphere for miles without
bumping into other particles. How does this affect the spread of air pollution?
And other researchers hope to study the nanogeoscience of Martian soil,
while still others want to develop computer simulations that model nanogeochemical
processes such as aggregation and sorption. Slowly, one study at a time,
they're unveiling the role of nanoscale oxide, sulfide, phosphate and
silicate particles in global phenomena.
"We need to study the unusual properties of natural materials at
the nanoscale," Waychunas says. "And we need to explore how
these properties relate to carbon sequestration, plant nutrient transfer,
toxic agent entrapment, and many other geochemical cycles."
They're off to a good start: for three days in mid-June, 85 researchers
from almost 30 universities, federal agencies, and national labs convened
at Berkeley Lab and mapped what is and isn't known in nanogeoscience.
In addition to bringing nanogeoscientists under one roof for the first
time, the workshop was tasked with helping federal agencies decide how
to best facilitate research. The attendees collated important, underfunded
research opportunities into a report that will be presented to several
federal agencies later this year.
Additional information:
|