|
 |
|
 |
|
|
 |
At the Advanced Light Source, robots
precisely mount tiny protein crystals in crystallography beamlines.
|
|
|
To learn more about life, Berkeley Lab researchers rely on robots. They've
automated a traditionally slow process in which tiny protein crystals
are mounted and centered in an x-ray beam and analyzed for their molecular
structure.
The robot, which is the first such device available to general users
at a synchrotron, both mounts protein crystals in a beamline and uses
the resulting data to decipher the protein's atomic makeup. Once fully
implemented, it will facilitate a ten-fold increase in the number of proteins
mapped annually at Berkeley Lab's Advanced Light Source (ALS), the world's
brightest source of ultraviolet and x-ray light. Such super-charged throughput
is needed to keep pace with the mountains of data produced by genome sequencing
and drug development research.
|
 |
|
|
|
|
|
At Beamline 5.0, researchers can determine the structure of proteins
from protein crystals only a few tens of millionths of a meter in
size. (Photo: NASA)
|
|
| |
|
"When it comes to synchrotrons, there is more demand than supply,"
says Thomas Earnest of Berkeley Lab's Physical Biosciences Division.
Synchrotrons work by accelerating electrons to nearly the speed of light
and bending them in a circular path by powerful magnets. At this speed,
electrons emit extremely bright x-ray light that is directed along a beamline
toward an object that researchers want to investigate at the atomic level
-- in this case protein crystals. The pattern in which x-rays diffract
off of the crystal is used to determine the protein's molecular structure,
which in turn is used to deduce the protein's function.
It's a laborious process. By hand, it takes roughly 10 to 20 minutes
to mount and align a protein crystal in a beamline. At this pace, it would
take years to muddle through the more than 30,000 proteins produced by
the human genome. People just aren't cut out for many of the repetitive,
round-the-clock tasks that comprise the protein-by-protein inquiry into
how genes function.
 |
|
|
|
|
|
 |
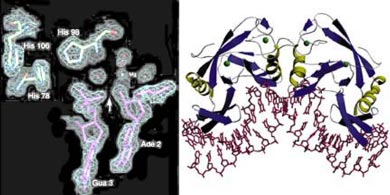
Electron density map of
a ribosomal structure determined at the ALS (Noller, UC Santa Cruz)
|
|
|
But machines love repetition. Earnest and colleagues' industrialized
approach to the data-acquisition phase of protein crystallography features
a container that houses 112 protein crystals. Nitrogen gas cryogenically
cools the crystals to about 100 Kelvin, an extremely low temperature that
must be maintained throughout the process in order to minimize radiation
damage.
Using a computer, an operator selects the crystals to be studied, and
a cryogenically cooled arm dips into the container, mounts a crystal on
a thin filament loop in about ten seconds, and centers it in the beamline.
A series of test exposures ensures that the crystal is both optimally
positioned and pure enough to provide reliable data. If everything checks
out, the robot obtains a full set of x-ray data. The entire process takes
only about two minutes per crystal -- and the result is enough data to
piece together a three-dimensional rendering of the protein's molecular
building blocks.
|
 |
|
| An enzyme's structural data from the
ALS (left) yields a diagram of its protein structure (right). (Stoddard,
Fred Hutchinson Cancer Center) |
|
The robot was developed by the Lab's Berkeley Center for Structural Biology
instrumentation group in collaboration with the Engineering Division's
bioinstrumentation group. It has been available to general users at ALS
beamline 5.0.3 since last summer and so far has helped analyze roughly
2,000 user-provided proteins. Earlier this year, a second robot was installed
on beamline 5.0.2, and two more are slated for installation in the near
future. In addition, Berkeley Lab researchers are helping to replicate
the system at Brookhaven National Lab's National Synchrotron Light Source
and Cornell University's High Energy Synchrotron Source.
In keeping with the robot's faster-is-better imperative, Earnest hopes
to soon add smart software that evaluates the data as it's collected,
and uses this feedback to constantly refine the data collection process
and how the crystals are chosen. This project is conducted in collaboration
with the Physical Biosciences Division's Computational Crystallography
Initiative.
In addition, crystal characteristics will be prescreened and fed to the
robot via a server. This data will enable the robot to more quickly zero
in on crystal-specific variables such as the optimal distance between
the crystal and the x-ray detector, and the angular range at which the
data is collected. This circumvents time-consuming trial-and-error steps
and further automates the process.
"The idea is crystals in, structures out," Earnest says. "Ultimately,
we would like to place 112 crystals in the container and walk away."
Another time-saver already in use is the so-called "hockey puck,"
a cylindrical cassette that holds 16 protein crystals. Seven pucks fit
inside a standard shipping container, and the same number can be nestled
in the robot's cryogenically cooled container. This means outside users
can prepare 112 protein samples and send them to the ALS, where they can
be quickly transferred to the robot for analysis.
Earnest cautions that automating the beamline phase of protein crystallography
isn't the only hurdle. Other bottlenecks remain, such as protein production
and crystallization. Even so, the robot is a critical component in the
push to increase the amount of structural information a beamline can produce,
he says.
In addition to basic research, the robot is also expected to help speed
drug development. Pharmaceutical companies are currently researching the
therapeutic effects of hundreds of enzymes and peptides that bind with
proteins. Understanding just how these compounds bind -- a process that
can be best visualized with x-ray diffraction -- will enable companies
to develop better pharmaceuticals. So the quicker companies can funnel
promising drug candidates through beamline analysis, the quicker they
can get them to consumers.
"Ultimately, we want to go from sequence to protein structure to
function, and the robot will help us to that," Earnest says. "This
saves the human brain for tasks it's better suited for, such as wondering
just how proteins work."
Additional information:
|