Mystery of Vital Cell Protein Solved After 30 Years |
 |
|
By Lynn Yarris, lcyarris@lbl.gov January 8, 1998 |
BERKELEY, CA -- A 30-year quest to solve the structure of one of the most important types of proteins in a living cell has been achieved. Scientists with the U.S. Department of Energy's Lawrence Berkeley National Laboratory have created the first 3-dimensional atomic model of tubulin, a protein that makes possible such vital life processes as cell division and the movement of materials within cells.
Eva Nogales, Sharon Wolf, and Kenneth Downing, biophysicists with Berkeley Lab's Life Sciences Division, announced the completion of their model in the January 8, 1998 issue of the scientific journal Nature. At a resolution of 3.7 angstroms, the model provides the first highly-detailed three-dimensional look at tubulin, including the site where the protein interacts with the promising anti-cancer drug taxol.
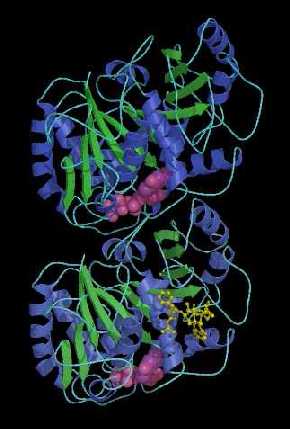
A ribbon diagram based on the 3.7 angstrom resolution model of tubulin developed by Berkeley Lab scientists shows the protein to be a dimer consisting of two monomers that are almost identical in structure. Each monomer is formed by a core of two beta sheets (blue and green) surrounded by helices, and each binds to a guanine nucleotide (pink). In addition to a nucleotide binding site, each monomer also has two other binding sites, one for proteins, and one for the anti-cancer drug taxol. | Download 1.8 Mg high-resolution TIF
Tubulin is the protein that polymerizes into long chains or filaments that form microtubules, hollow fibers which serve as a skeletal system for living cells. Microtubules have the ability to shift through various formations which is what enables a cell to undergo mitosis or to regulate intracellular transport. The formation-shifting of microtubules is made possible by the flexibility of tubulin which is why scientists have sought to understand the protein's atomic structure since its discovery in the 1950s.
Interest in tubulin structure heated up intensely in recent years when taxol, a natural substance found in the bark of the Pacific yew tree (the name "taxol" has been trademarked by Bristol-Myers-Squibb), was shown in clinical tests to be an effective treatment for a number of cancers including ovarian, breast, and lung. Cancer occurs when cell division runs amok.
By binding to tubulin and causing the protein to lose its flexibility, taxol prevents a cell from dividing. With better knowledge of tubulin structure and its interaction with taxol, scientists believe that an even more effective anti-cancer drug, one that interacts only with the tubulin of cancerous cells, could be synthesized.
"An awful lot of people have wanted to know what tubulin looks like at the atomic level," says Nogales. "The knowledge gained from this model should be of invaluable help in understanding the microtubule system in the cell."
Tubulin is a "heterodimer" protein, meaning it is comprised of a pair of polypeptide chains -- called monomers -- that differ in the sequence of their amino acids. The model presented by Nogales, Wolf, and Downing, shows that each tubulin monomer -- the alpha and the beta -- is a compact molecular structure with three functional components or domains, one that binds to nucleotides, one that binds to drugs like taxol, and one that looks to be a binding site for other proteins.
"The interaction between domains is very tight so that the effects that nucleotides, drugs, and other proteins have on tubulin are firmly linked," says Nogales. "The assembly of tubulin and its regulation through dynamic instability results from the fine tuning of the three components."
To produce their model, the Berkeley Lab researchers first polymerized tubulin proteins under the same conditions in which microtubules are formed except for the additional presence of zinc. The zinc prevents tubulin chains from curling around into hollow fibers. Instead, the polymerized tubulin forms two-dimensional crystalline sheets that are ideal for imaging by electron crystallography. The use of electron-based rather than the x-ray-based crystallography techniques customarily employed in protein studies was crucial to the model's 3.7 angstrom resolution.
"Working with x-ray crystallography requires much more protein than electron crystallography," says Downing. "Obtaining diffraction patterns with an electron beam enables us to work with crystals only one molecule in thickness, giving us our high resolution."
The final 3-D model is a computerized reconstruction derived from a data set that included 93 electron diffraction patterns and 159 images culled by the researchers from the more than 4,000 images which were recorded over a six-year period. The diffraction patterns and images were generated on an electron microscope equipped with a special "cold stage" that reduced damage to the crystals from the electron beam and yielded less "noise" than a conventional electron microscope. This microscope is also equipped to allow the tilting of samples at various angles so images can be obtained from different directions.
"We needed to keep our samples extremely stable during the imaging process in order to get the resolution we wanted," says Nogales. To this end, she and her colleagues used taxol to lock their samples into fixed positions for imaging. That the taxol would bind to their crystals is in part a confirmation that their structural model of tubulin is accurate.
The Berkeley Lab is a U.S. Department of Energy national laboratory located in Berkeley, California. It conducts unclassified scientific research and is managed by the University of California.