
Finding
an Alzheimer’s Switch
Three Lab Scientists
Elected to the NAS
Finding an Alzheimer’s Switch
An Unsuspected Protein Regulates the Production of Plaque-Forming Peptides
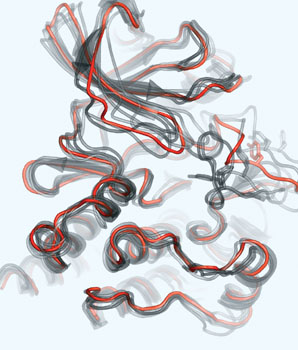
Life Sciences Division researchers have discovered a key player in Alzheimer’s disease, an unsuspected subunit of the gamma-secretase enzyme. They found that the protein CD147 regulates the production of toxic peptides that cause amyloid plaques — Alzheimer’s defining brain lesions.

Left to right, scientists Hua Zhou, Peter Walian, Bing Jap, and Shuxia
Zhou made a key discovery in Alzheimer research.
“Alzheimer’s is worse than a disease — it takes the soul of a human being,” says Bing Jap, who directed the research. “As the population of this country ages, the incidence of Alzheimer’s is increasing.” Jap and his colleagues Shuxia Zhou, Hua Zhou and Peter Walian report their findings in the early edition of Proceedings of the National Academy of Sciences, now online.
Alzheimer’s invades the brain after a protein called amyloid precursor protein (APP) is clipped into shorter pieces by enzymes known as secretases. If the portion of APP clipped by the beta form of secretase is further clipped by a third form, gamma-secretase, the resulting fragments are amyloid beta-peptides, which form amyloid plaques.
APP and secretases are membrane proteins in the brain’s neural cells; unlike most such proteins, gamma-secretase acts within the thin membrane to make its crucial cut. Research would greatly benefit if its high-resolution structure could be established by x-ray crystallography.
“It’s extremely difficult to get enough pure membrane protein to crystallize,” says Jap, whose laboratory has a reputation for solving the structures of important membrane proteins. As the first step to producing enough gamma-secretase to make crystals, Jap asked postdoctoral fellow Shuxia Zhou to characterize the native protein complex.
“Previous experiments establishing the role of gamma-secretase were genetic experiments done by causing its overexpression in cell lines and animal models,” Zhou explains. “We wanted to isolate the native form and purify the whole complex.” The researchers isolated the native complex from cells of the HeLa line, then pulled its subunits apart by gel electrophoresis.
“There were six strong bands in the gel, five of which we could identify,” she says. These represented the four known subunits of gamma-secretase, one of which is divided in two parts in the mature complex. Previous experiments had showed that a complex of these four components was enzymatically active, but apparently this was not the native form.
“In addition to these five bands we found an extra band,” Zhou says. “We didn’t know what it was.” She and her colleagues clipped the band from the gel, extracted the protein, and sequenced its amino acids. The mystery protein turned out to be CD147.

CD147 was familiar, expressed in many tissues and having many biological functions, including neural functions. When the CD147 gene is deleted in mice, the results include loss of memory and disorientation, suggestive of Alzheimer’s disease.
To investigate CD147’s part in the activity of gamma-secretase, the researchers used targeted RNA to silence CD147 in cell cultures. When CD147 was silenced, production of amyloid beta-peptides increased markedly.
Although CD147 appears in other cell lines and in many contexts besides gamma-secretase, only as a part of gamma-secretase does it regulate the production of amyloid beta-peptides and thus amyloid plaques. How does it do its normal job of preventing excessive production of the toxic peptides, and what causes it to fail?
“We know CD147 is a regulatory subunit of gamma-secretase, but we don’t know how it works,” Zhou says, “Determining this mode of action is a key goal of our future efforts. The answer has to wait for high-resolution structural work, and for that we first have to make enough of the native complex to make crystals.”
Says Bing Jap, “Determining the atomic structure of the gamma-secretase complex, including CD147, is the next crucial step to designing Alzheimer’s disease therapeutics.”
Three Lab Scientists Elected to the NAS
Three distinguished Berkeley Lab scientists were among this year’s 72 newly-elected members of the National Academy of Sciences (NAS). Carolyn Bertozzi, Steve Louie, and Barbara Romanowicz have joined the 1,976 active members of NAS. Membership in this organization, founded by Abraham Lincoln in 1863, is considered one of the nation’s most prestigious honors for a scientist or engineer.
Carolyn Bertozzi

Carolyn Bertozzi
A member of Berkeley Lab’s Materials Sciences and Physical Biosciences divisions, Carolyn Bertozzi is also a professor of chemistry, biochemistry, and molecular biology at UC Berkeley and director of the Biological Nanostructures facility at the Molecular Foundry.
Bertozzi combined her knowledge of physical and synthetic-organic chemistry with experience in medicine and immunology to create a new field of research — the design and engineering of cell surfaces by programming expression of complex molecules not found in nature. Among the potential applications are targeting tumor cells and other disease cells for diagnosis and treatment, inserting genes into specially labeled cells, and designing “cyborg” cells joined with artificial materials. Less than a year ago, following a series of demonstrations of cell-surface engineering in vitro, Bertozzi and her colleagues reported the remodeling of cells in living mice, the first time cells in a higher animal had been synthetically modified to undergo chemical reactions without physiological harm.
Bertozzi’s numerous awards include the MacArthur Foundation “genius” Fellowship (given when she was 33 years old), appointment as a Howard Hughes Medical Institute Investigator, and teaching recognitions including UC Berkeley’s Distinguished Teaching Award and the Donald Sterling Noyce Prize for Excellence in Undergraduate Teaching. She’s also an occasional rock musician.
Steven Louie

Steven Louie
Steven Louie is a member of the Lab’s Materials Sciences Division, a professor of physics at UC Berkeley, and director of the Theory of Nano-structured Materials at the Molecular Foundry. His many honors include the 2003 Richard P. Feynman Prize in Molecular Nanotechnology from the Foresight Institute (shared with his frequent collaborator Marvin Cohen). According to the Information Sciences Institute, Louie is one of the 25 most highly cited authors in nanoscience and one of the 100 most cited researchers in all of physics.
Louie’s role as a pioneering theorist began when his graduate studies at UC Berkeley coincided with Berkeley Lab’s acquisition of a state-of-the-art “supercomputer,” a Control Data 7600; he has been a leader in computational physics ever since. His theoretical investigations include the electronic and optical properties of numerous nanotube compositions and configurations, the design of carbon nanotube diodes, and prediction of fullerene properties.
Louie and his colleagues have solved puzzles like the anomalous double energy gap of magnesium diboride, a remarkable high-temperature superconductor that is not a ceramic but a metal, and the mystery of the Earth’s “missing xenon,” once thought to have been captured by the molten iron core, a hypothesis Louie disproved with computer modeling.
Barbara Romanowicz

Barbara Romanowicz
Barbara Romanowicz joined Berkeley Lab’s Earth Sciences Division as an associate staff scientist last year. Since 1991, she has been a professor of geology and geophysics at UC Berkeley and the director of the Berkeley Seismological Laboratory. In 2002, she became chair of UC Berkeley’s Department of Earth and Planetary Science.
Her research focuses on the structure and dynamics of the deep earth, from the crust to the inner core. She also studies global seismic events and their relation to plate tectonics, as well as global earthquake scaling laws. Her group tackles theoretical wave propagation problems in complex three-dimensional media, and tomographic computer modeling of Earth’s mantle.
Recently, Romanowicz co-authored a paper, published in the journal Nature in 2004, which explains the source of mysterious waves that constantly reverberate through the Earth, even on days when earthquakes don’t occur. She found that the oscillations originate from storm-driven sea waves that interact with one another and reach deep into the ocean. In another paper, published in the journal Science last month, she and two other scientists looked at seismic waves that traverse through the Earth’s inner and outer core, and confirmed that the Earth’s core is indeed solid.
Eighth Graders Learn About Solar Neutrinos
 Kevin
Lesko
Kevin
Lesko
Kevin Lesko of the Nuclear Science Division was among the scientists who presented lectures to 44 eighth graders from Oakland’s Redwood Day School last week. Lesko, who has organized such presentations for the past six years, discussed the use of solar neutrinos in underground experiments.
“The idea is to expose the children to cutting edge science — not the textbook version, but what will be in the text books in a few years — and for them to meet some of the people involved in the research. In the process we also give them an impression of what Berkeley Lab is all about.”
The students also heard from nuclear scientists Charles Currat, Gersende Prior, and Reyco Henning and toured the Advanced Light Source.
“Each of these visits to the laboratory has sparked a real interest in one or two of the students,” Lesko said, “enough so that several of them come up afterwards to ask more questions, sometimes weeks later.”
Books Bound for Iraq Thanks to JGI Researcher
In distant lands torn by war, packages of biology and chemistry textbooks, classic English novels, and dictionaries are arriving every month from the United States to promote education and to restock the shelves depleted by prolonged conflict.

JGI researcher Bob Macey points out the location in Northern Iraq
where the next shipment of books will be sent.
These packages come care of Joint Genome Institute (JGI) scientist Bob Macey. He and his colleagues at the California Academy of Sciences, the Smithsonian Institution, and the American Museum of Natural History in New York are partnering to provide reading materials to such far-flung places in need as Iraq, Afghanistan and Sudan.
The “Better World Through Books” program evolved serendipitously through Macey’s investigations into the genetics of Middle Eastern lizards. “While working on a National Science Foundation grant, I enlisted the services of USAID to help establish contacts with scientists in Iraq,” Macey said. “I also happened to mention that we have helped coordinate book drives, rounding up libraries of used and new books to send to our colleagues in the lesser developed world. Two hours later I received a call back from the head of USAID in Erbil saying they’d be willing to assist in distributing the books.”
In February, the first book shipment — some 450 pounds — was shipped out to USAID in Kurdistan, the far northern reaches of Iraq, for delivery to Salahaddin University in Erbil. Since then, an additional 700 pounds have been shipped to Erbil. In response, the University has been able to establish a new biology library. Since then, the program in Iraq has reached out to Mosul and points beyond.
Macey took his cue from UC Berkeley researcher Ted Papenfuss at the Museum of Vertebrate Zoology, who pioneered the book program in Somalia and Sierra Leone over the last two years. In Somalia, Papenfuss said, after they conduct a class, each student is given a single book to take with them on their teaching assignment in the hinterlands, so the influence is spreading.
To date, some 28 boxes with more than a ton of books have been sent to Iraq alone. Among the most notable contributors to the book program is Kraig Adler from Cornell University and the Society for the Study of Amphibians and Reptiles, who have donated several thousand dollars’ worth of new texts and journals targeted for Iraq and Afghanistan. Jonathan Losos of Washington University in St. Louis has in turn shipped over 600 pounds of books received at Iraqi Univer-sities in Erbil, Mosul and Sulaymani, and bound for Afghanistan.
“I’m really excited at the momentum we are building for ‘A Better World Through Books,’” Macey said. “I invite everyone to join the effort.” They seek books — anything educational, in any language. Old books are not necessarily bad. “More,” is the operative word. “It is hard for those who haven’t been to these remote places to appreciate how little you have to contribute to make such a huge difference.”
Papenfuss reported that at Amoud University in Somalia, they only had one biology book to teach with at an entire university. That was before the book program took off. Now they have 200.
“Collectively we have decided to effect this change across 40 countries,” Macey said. He said that spring is the perfect time for purging your bookshelves and donating to this worthy cause.
Book donations can be sent to Bob Macey, care of JGI-PGF, L-450 or c/o the Museum of Vertebrate Zoology, 3101 Valley Life Science Building, University of California, Berkeley, California, 94720.
Protein Dynamics on the Supercomputer Big Screen
Now playing at a supercomputer near you: proteins in action. Scien-tists from Berkeley Lab and UC Berkeley are using one the world’s most powerful computers to simulate how protein molecules move, rotate, and fold as they carry out life’s most fundamental tasks.
 This simulation of a tyrosine kinase reveals how the protein changes shape.
This simulation of a tyrosine kinase reveals how the protein changes shape.
Although they only approximate real-life phenomena, the increasingly realistic movies are becoming useful complements to real-world experiments in helping scientists determine how proteins function. Using them, biologists can gain a better understanding of how incorrectly folded proteins lead to a range of diseases, or how other proteins synthesize adenosine triphosphate (ATP), the fuel that powers many biomolecular motors.
“Proteins are very complex molecules with thousands of atoms, but they don’t come with a user’s manual,” says John Kuriyan of Berkeley Lab’s Physical Biosciences Division. “Fortunately, over the past few years, rapid increases in computing power and better simulation programs have made it possible to visualize protein dynamics like never before.”
The simulations are created at the National Energy Research Scientific Computing Center (NERSC), which is located at Berkeley Lab and is the flagship scientific computing facility for DOE’s Office of Science. NERSC boasts the raw power needed to develop simulations that are detailed enough to capture a protein’s fastest movement, and long enough to portray their relatively infrequent but biologically important changes. In some cases, this means stringing together femtosecond-length, atom-scale snapshots of a 50,000-atom protein, frame by frame, into movies that span several nanoseconds. (A femtosecond is one-millionth of a nanosecond, and a nanosecond is one-billionth of a second.)
Although these high-resolution simulations take days to prepare even on a supercomputer, they enable Kuriyan and colleagues to test-drive proteins under a variety of conditions. They can see what happens when a protein is given just enough energy to teeter on the edge of a conformational change. Or they can prod a protein to change shape, and gauge how forcefully it resists or how readily it gives in.
“The simulations allow us to push here and there and determine how the protein responds,” says Kuriyan, who is also a Howard Hughes Medical Institute investigator and a Chancellor’s Professor in UC Berkeley’s Department of Molecular and Cell Biology and Department of Chemistry. “This is important because it isn’t always obvious which experiments will address a protein’s mechanistic properties.”
Berkeley Lab researchers aren’t the only scientists spearheading the development of these virtual protein movies, but they’re uniquely suited to lead the way. Along with NERSC, Berkeley Lab and UC Berkeley have joint appointees like theoretical chemists David Chandler and Phillip Geissler, who are constantly refining the fundamental molecular theories on which the simulations are based. Add the expertise of experimental biologists like Kuriyan, who put real proteins through the wringer to learn how they work, and the Lab has an ideal blend of theory, practical know-how, and computing power to create almost lifelike movies.
So far, Kuriyan and colleagues have used NERSC simulations to learn how certain proteins, called Src tyrosine kinases, transmit signals initiated by growth factor receptors in human cells. Mutant forms of these proteins can trigger cancer. They’ve also simulated the conformational and energy changes that proteins involved in DNA replication must undergo in order to rapidly copy DNA strands.
In each case, the simulations furthered their understanding of protein dynamics and helped guide real-world experiments. They also underscored the need for powerful computers. The quickest motion in a protein is the stretch of the carbon-hydrogen bond, which occurs in about one femtosecond. This means that each frame of a simulation must depict a protein’s movement femtosecond by femtosecond. If it doesn’t, the simulation will skip over these carbon-hydrogen stretches and be as true to life as a jerky 1920s movie. But interesting changes in proteins, such as the rotation of a portion of the ATP-making enzyme, often occur in the microsecond to millisecond time scale — up to nine orders of scale slower than a femtosecond. In other words, a simulation must weave together billions of femtosecond-length snapshots in order to capture one or two rare but important changes.
The dilemma is like filming the muscular movements of a person. To capture the smallest muscle twitch, down to a single heartbeat, the film must have several frames per second. Unfortunately, the person might not do something significant, like go skydiving, for days. The filmmaker must churn through miles of film reel in order to record that infrequent but important leap from an airplane.
“This is why computer speed becomes very important. The faster the computer can simulate each frame of the movie, the more frames can be generated, and the sooner we will get to something interesting,” says Kuriyan.
He adds that the simulations aren’t perfect. They’re constructed frame by frame, so no matter how fast they become, they will always gloss over some nuance of a protein’s motion. In addition, a ten-nanosecond simulation of a large protein molecule sometimes requires 20 to 40 days of a supercomputer’s processor time to create.
“We’d like to run a simulation and get the answer in a day, so we can change what we are doing in the lab,” says Kuriyan. “We’re not close to being there yet. But increases in computer speed have enabled dramatic advances recently, and this trend will continue. It is very important to support national resources like NERSC that help maintain competitiveness and very fast computation.”
Kuriyan and Martin Karplus of Harvard University discuss the promise of protein molecule simulations in a paper entitled “Molecular dynamics and protein function,” which was published online by the Proceedings of the National Academy of Science on May 3.
Retired But Not Forgotten, Shank Will Get His Day
When he retired after 15 successful years as Berkeley Lab’s director last year, Chuck Shank made a quiet exit, shunning the fanfare and the traditional farewell ceremonies. But many people, including Distinguished Scientist Mina Bissell, felt this modest departure and this man didn’t get nearly the attention they deserved.

Charles Shank
“Chuck has a well-earned reputation as one of the great visionary directors of Berkeley Lab,” Bissell said. “He is an exceptional leader who oversaw remarkable growth and progress and brought the Lab to the forefront of modern scientific research, achievement and excellence.”
So she chaired an organizing committee that put together an impressive all-day symposium to pay tribute to Shank’s management and scientific distinctions. On May 24 in the Building 50 auditorium, a series of 30-minute presentations by some of science’s most respected practitioners will address “Science of the 21st Century” and honor the person whose efforts encouraged much of it.
The special guests include John Marburger, science advisor to President Bush; Robert Birgeneau, chancellor of UC Berkeley; Gerry Rubin, director of the Janelia Farm Research Campus of the National Institutes of Health; and two award-winning scientists: chemist Nate Lewis of Caltech and physicist Erich Ippen of the Massachusetts Institute of Technology. Home-grown talent making presentations include mathematician Phil Colella, biophysicist Eva Nogales, geochemist Jill Banfield, and astrophysicist Saul Perlmutter.
Former Deputy Director Pier Oddone, who will be Fermilab’s next director, and Bissell will serve as hosts, and current Berkeley Lab Director Steve Chu will offer the first talk, “What Can Physicists Learn from Biology?”
Marburger, who heads the U.S. Office of Science and Technology Policy and was once Shank’s director colleague at Brookhaven National Laboratory, will give the lunch keynote address, entitled “There’s Plenty of Time at the Bottom.” Birgeneau, who like Ippen and Chu were former Shank colleagues at Bell Laboratories in New Jersey, will follow with a discussion of “Liquids, Crystals, and Liquid Crystals.”
The $50 registration fee includes all sessions, lunch and a reception. To reserve a spot call Kim Magbie at X7008. The full agenda can be viewed at the symposium website, www.lbl.gov/DIR/symposia/.
During his return to Berkeley, Shank will also deliver the commencement address for the College of Chemistry on campus.
A Phase Transition in a League of its Own
There are quick-change artists and there is vanadium dioxide. Scientists knew the material was fast at making the change from electrical insulator to conductor, but they had no idea just how fast. Berkeley Lab scientists have now clocked the transition time at shorter than 150 femtoseconds (about one-tenth of a trillionth of a second), which is slightly less than the time it takes a beam of light traveling at 300,000 kilometers per second to cover a distance of one-tenth of a millimeter.
“This is certainly the fastest solid-to-solid phase transition ever observed,” said Andrea Cavalleri, a physicist in the Materials Sciences Division (MSD) who has been leading the vanadium dioxide effort. “Such a fast phase change could be very useful for optical devices, particularly for high-speed switching.”
 Matteo Rini (left) and Andrea Cavalleri used femtosecond spectroscopy
techniques to measure the speed at which thin films of vanadium dioxide
make the phase transition from an electrical insulator to a conductor.
The speed was clocked at about one-tenth of a trillionth of a second.
Matteo Rini (left) and Andrea Cavalleri used femtosecond spectroscopy
techniques to measure the speed at which thin films of vanadium dioxide
make the phase transition from an electrical insulator to a conductor.
The speed was clocked at about one-tenth of a trillionth of a second.
Cavalleri, winner of a European Young Investigator Award for his ultrafast studies of complex materials, is a member of the femtosecond spectroscopy group of Robert Schoenlein. He, Schoenlein and other members of the group have been conducting ongoing investigations into the superfast insulator-to-metal transition in thin films of vanadium dioxide. The ability of vanadium dioxide, a non-magnetic semiconductor, to change from a transparent insulator to a reflective conductor was first reported back in 1959, but the speed at which the phase transition takes place was not known until Cavalleri began his studies while at UC San Diego.
“Vanadium dioxide is a strongly correlated compound (a class of materials that includes high-temperature superconductors and ferroelectrics), which means there’s a strong relationship between structural and electronic effects,” said Cavalleri. “Because structural and electronic effects in these materials change almost simultaneously across a phase transition, we were never able to assess which was driving which. The mystery had been compared to the question of which came first, the chicken or the egg.”
In one series of studies at Berkeley Lab, thin films (about 50 nanometers in thickness) of vanadium dioxide on silicon wafers, with a thin layer (about 200 nanometers) of silicon nitride as a buffer, were zapped with 100 femtosecond pulses of white laser light. This not only triggered the phase transition of insulator to metal, but also resulted in a host of subtle structural changes that were observed through time-resolved spectroscopy techniques.
Said Schoenlein, “The transition time for the phase change was determined by femtosecond measurements of changes in the material’s optical absorbance. Funda-mental physics studies were done via femtosecond measurements of reflectivity changes.”
As reported in the journal Physical Review B, Cavalleri, Schoenlein and their colleagues found evidence that pointed to the “chicken” coming first. That is, the phase transition in the vanadium dioxide results from a structural rearrangement of the atoms in the crystal lattice. The big surprise was that this atomic motion was coherent, as opposed to the random motion of atoms in a crystal that occurs during thermal heating.
Explained Schoenlein, “In vanadium dioxide, the phase change can be driven on the femtosecond time scale directly by the electron-hole pairs that are created when photons are absorbed. Eventually the energy of these carriers is transferred to the lattice in the form of heat, but the phase transition we are observing occurs long before that. It turns out that the limiting time scale — approximately 70 femtoseconds — corresponds to a specific motion of the atoms in the lattice, and strongly indicates that the atoms must be moving in a coherent way.”
Added Cavalleri, “We have found that if you excite the electrons out of their ground state while the atoms in the lattice are still frozen in place, a process similar to chemical doping, it is still not enough to make the vanadium dioxide thin film metallic because you have to wait for the atoms in the lattice to move. Specifically, it is the motion of the vanadium atoms that’s the bottleneck to the formation of the metallic phase.”
There was some speculation that it was not a complete phase transition that was being measured, but rather a transition from an insulator to an intermediate phase, as is common in other semiconductors. In their most recent results, however, reported in the journal Optics Letters, the Berkeley Lab researcher confirmed they were measuring a complete transition from insulator to conductor through the observation of a special electron wave called a “surface plasmon resonance,” which only occurs on the surfaces of metals.
In the experiments reported in Optics Letters, the ultrafast optical measurements were carried out by Matteo Rini, another member of Schoenlein’s group, working with samples of vanadium dioxide nanocrystals that were prepared and characterized by René López, a member of the research group of Richard Haglund at Vanderbilt University. These results showed that nanocrystals provide some degree of tunability and enhancement of vanadium dioxide’s optical properties after the phase transition.
Vanadium dioxide’s superfast insulator-to-metal transition bodes well for future high-speed optical switches and other devices. For example, being a solid-to-solid phase transition, the process is reversible. Furthermore, the phase transition can be initiated with just a tiny amount of energy, which is ideal for optical switching.
“Our studies are showing the way for the use of vanadium dioxide, both as thin films and as nanoscale particles, in a number of new applications in photonics,” said Cavalleri. “But there are a lot of other oxides of transition metals that may ultimately be even better.”
Celebrating a Miraculous Century of Physics
A Conversation with James Siegrist
Around the globe, hundreds of events are scheduled to celebrate the World Year of Physics 2005. In the U.S., these include everything from sober scientific symposia to pumpkin-catapulting contests. Berkeley Lab will sponsor a series of events, on the Hill and off, aimed at conveying the relevance and excitement of physics to students and the public.
To find out more about this physics fête and Berkeley Lab’s participation, the View spoke with Jim Siegrist, director of the Physics Division and associate laboratory director for General Sciences.
View: Why was 2005 chosen as the Year of Physics — what are we celebrating and why?
Siegrist: In 1905 Einstein published three great papers: one on Brownian motion, which in effect proved that atoms exist; one on special relativity; and one on the photoelectric effect, which established the quantum nature of light. Historians call it Einstein’s “miracle year.” The centennial is a good chance for public outreach and to show that physics is actually useful to society.
View: Playing devil’s advocate, just how is physics useful to society?
Siegrist: Ray Orbach (director of DOE’s Office of Science) lists three reasons. One is economic growth through technical advances, including applications of unexpected discoveries — for example, who would ever have thought that by working on particle physics you’d invent the worldwide web? You can’t get much more serendipitous than that. Orbach also names scientific literacy, which supports economic prosperity through a
better-educated, more productive workforce. And the third is intellectual curiosity, the fundamental questions about how the universe began and how the world works.
View: Physics surely looks a lot different today than it did 100 years ago.
Siegrist: Yes and no. The photoelectric effect is one of the basic things you study on your way to figuring out quantum mechanics — the solid-state-physics applications of quantum mechanics are certainly important to the Bay Area, because we wouldn’t have the semiconductor industry unless we had figured out how to tame silicon atoms. So in those respects today is different from 1905. In other ways, however, Einstein was personally consumed with trying to reconcile general relativity and electrodynamics, which he never succeeded in doing, and which we still haven’t succeeded in doing, really. Connecting general relativity and quantum mechanics remains the main big question.
View: How are these vastly different scales connected — the microworld of subatomic particles and the cosmos?
Siegrist: In Einstein’s time, and for a long time after, these were parallel tracks; it used to be that the way you studied particle physics was with cosmic rays. Then, with accelerators right after the war, nuclear physics and particle physics grew away from astrophysics. But recently, because of all the dark energy and dark matter discoveries, they’ve come back together again.
View: Does that mean accelerator studies and astrophysics are really looking at the same thing?
Siegrist: We don’t know if it’s exactly same thing, but we know there’s a problem with understanding the vacuum. The Large Hadron Collider is going to look at how the vacuum ended up generating the mass of the particles. And from all the studies of the last 10 years we know that the dark matter is particles. It’s not Jupiters that are just hidden from view; in some sense it’s ashes left over from the Big Bang. The lightest supersymmetric particles could be the bulk of the dark matter, but there are other models. Visible matter is a complicated mixture of stuff, so it’s hard to believe the dark matter is just one thing. The LHC, and the International Linear Collider after that, should be able to untangle the properties of those particles, which will bring a closure between the particle physics side and the astrophysics side. To do that we’ll eventually have to do what Einstein set out to do, which requires bringing quantum mechanics and gravity together.
View: How will Berkeley Lab contribute in the future?
Physics Division Director James Siegrist in front of a SNAP model.
Siegrist: We’re working on LHC magnet technology and the tracking detectors for ATLAS, and we hope to follow with detector and accelerator work for the International Linear Collider, which we’re just starting.
On the astrophysics side, we hope SNAP will get flown as the Joint Dark Energy Mission. The nature of dark energy is one of the most important questions physics faces in the 21st century — it has been called the most fundamentally mysterious thing in basic science — and it was discovered because of the supernova work initiated and followed through here at Berkeley Lab. We're also planning experiments to measure the polarization of the cosmic microwave background radiation, in the R&D phase right now, which will let us actually see the effects of gravitational waves from the first moments of inflation.
Another future project is the Rare Isotope Accelerator — we’re working on ion sources and detectors — which will address the rare nuclear processes that produced the elements beyond iron. And the other front we’re on is “neutrino land.” We’re involved in a range of collaborative programs addressing many questions — everything from IceCube at the South Pole, studying cosmic neutrinos, to asking whether the neutrino is its own antiparticle, and whether there is CP violation in the neutrino sector. We’ve found CP violation in the quark sector, but it can’t account for all the missing antimatter in the universe. What you hope for is large enough CP violation that you can explain where all the antimatter went.
View: So the challenge of the program World Year of Physics is to address fundamental questions and at the same time explain how physics is useful to society.
Siegrist: Robert Nadeau will talk about the physics of earthquakes, one of the things we care about around here. The spinoff work by Carl Haber, using optical techniques from particle physics to reconstruct historic audio recordings — people find that pretty exciting. Steve Chu will talk about trying to solve the energy problem by converting solar energy to chemical energy; Berkeley Lab brings strength in both physics and biology to the hope of copying what biological systems do, only better. As for fundamental questions, Saul Perlmutter will talk about dark energy, and in October we’ll be hosting a symposium on the 50th anniversary of the discovery of the antiproton, the p-bar, which proved that antimatter is real and the posi-tron wasn’t just some fluke. A whole boatload of physics flowed out of that discovery. And there’s tons more.
More information on Berkeley Lab events for the World Year of Physics will appear in future issues of the View and Today at Berkeley Lab. Visit the World Year of Physics website at www.physics2005.org/.
Engineering’s Composite Capabilities
Before joining Berkeley Lab’s Engineering Division to work on the Physics Division’s ATLAS Pixel Detector, Neal Hartman and his supervisor, Eric Anderssen, had both gained experience working with an increasingly important class of materials known as composites. From satellites to bicycles — or Anderssen’s speciality, “human force amplifiers” (think of Sigourney Weaver duking it out with the bug queen in Aliens) — anything requiring stiffness and minimal weight is a candidate for modern composite construction.
 ATLAS team members (left to right) Jon Wirth, Tom Johnson, Mario Cepeda, Alexis Smith, Neal Hartman, and Eric Andersson are pictured with the support structure for ATLAS' Pixel Detector.
ATLAS team members (left to right) Jon Wirth, Tom Johnson, Mario Cepeda, Alexis Smith, Neal Hartman, and Eric Andersson are pictured with the support structure for ATLAS' Pixel Detector.
Composites go back at least 5,000 years, to the first Egyptian sheets of papyrus, but these days the term usually indicates fibers like carbon or glass embedded in a matrix like epoxy resin; such composites can be up to 10 times stiffer than metal alloys by weight. For some applications, composites are the only logical choice.
The framework for the ATLAS Pixel Detector is an example. After it is slid into place at CERN, the structure will have to maintain virtually perfect alignment over its entire 7.5-meter length (including service-support modules). To resist intense radiation and not interfere with particle paths, its mass must be minimal.
“When I got here, the main support frame for the detector itself had already been made offsite, because we didn’t have the capability to make composites at LBL,” says Hartman. Several factors indicated it would be cost effective to make the rest of the parts at Berkeley Lab. “People were initially skeptical about how much risk was involved, but Eric and I were strong proponents of developing this capability, which could be passed down to newer projects.”
Modern composites come from the manufacturer in cloth-like rolls impregnated with resin. Typically several layers of material are built up on a metal mold, then baked in an autoclave, which applies pressure to squeeze out excess resin. With help from Pixel Project funds, the Engineering Division’s Mechanical Engineering Department acquired a walk-in cold storage unit, needed to keep raw materials from setting prematurely; an automated fabric cutter to cut the layers into shape; and a 10-foot-long autoclave capable of temperatures of 450ºF (222ºC) and up to 10 times atmospheric pressure.
The trickiest step in making a composite part, says Anderssen, is calculating how it is going to expand as it cures. Despite sophisticated programs that take into account factors like the shape, composition and orientation of the different layers, some trial and error is inevitable. This is the most expensive part of the process; after the first half-dozen or so prototypes, individual pieces can become “dirt cheap.”
Good record-keeping is essential. “All the information for each part is stored in a database, accessed by a bar code on the part,” Anderssen explains, “including where its raw materials came from, their age, and even the pressure and temperature measured by sensors on the part during autoclaving.”
The successful completion of the ATLAS Pixel Detector support structures, and the first detector disk with sensors in place, is an indication that the Engineering Division is prepared to produce an enormous range of composite structures of high quality and stringent accuracy — thereby “opening the door,” says Hartman, “for many future opportunities.”
Paper Sensor Passes Major Milestone
The paper industry is one step closer to saving millions of dollars each year. An innovative laser ultrasonic sensor designed and built by Berkeley Lab scientists was recently successfully tested at a paper mill in Jackson, Alabama.
 The laser ultrasonic sensor was successfully tested at a working paper
mill.
The laser ultrasonic sensor was successfully tested at a working paper
mill.
The sensor measures a paper’s bending stiffness and shear strength — two hallmarks of paper quality — as it speeds through a production web. By doing so, it can ensure that the optimum amount of raw material is used to make the paper, which could reduce the consumption of trees and chemicals and save the U.S. approximately $200 million in energy costs and $330 million in fiber costs each year.
“This is the first full-scale demonstration of the sensor on a commercial paper-making machine while it’s in operation,” says Paul Ridgway of the Environmental Energy Technologies Division (EETD), who developed the sensor with the project’s principal investigator and fellow EETD scientist Rick Russo. “Mill engineers considered the trial to be quite successful, and are hopeful that a six-month trial will be conducted at the same mill.”
The two-week test, conducted in February at a mill owned by Boise Cascade, also included experts from Georgia Tech’s Institute of Paper Science and Technology, which collaborated with Berkeley Lab on the project. Eight years in the making, the sensor was funded by the Department of Energy’s Office of Industrial Technologies as part of a partnership to improve the energy efficiency of several industries. Under this program, the American Forest and Paper Association created Agenda 2020, which outlines ways in which the forest products industry will streamline its production processes.
Papermaking is an obvious candidate for improvement. To gauge paper quality today, a 15-to-30-ton paper roll is manufactured, and then a small sample is obtained and analyzed for its mechanical properties by observing how it bends. If the sample doesn’t meet certain specifications, the entire roll is recycled into pulp or sold as an inferior grade. To avoid this costly mistake, manufacturers often over-engineer paper and use more pulp than necessary to ensure the final product isn’t substandard. This method consumes more raw material and energy than necessary, so the Berkeley Lab team developed a sensor that tracks paper’s flexibility on the fly — in real time. Speci-fically, the sensor measures the time it takes ultrasonic shock waves to propagate from a laser-induced excitation point on the moving paper to a detection point several millimeters away. The velocity at which the ultrasound waves travel from the excitation point through the paper to the detection point is related to two elastic properties, bending stiffness and out-of-plane shear rigidity.
The laser ultrasonic sensor conducts these measurements without touching the paper, an important advantage given that the paper moves at 20 meters per second (45 miles per hour) and the slightest contact can mar lightweight grades such as copy paper and newsprint. The recent trial also boasted the highest sample speed ever reported for a commercial application of laser ultrasonics.
The next step in the project is to work with Boise Cascade to link the sensor with sophisticated feedback controls that maintain the paper’s stiffness while it’s being manufactured. ABB Corporation, which participated in the recent trial, is also likely to participate in this phase.
“Our technology will enable this real-time feedback control,” says Ridgway. “And the successful mill trial shows we are one step closer to realizing it.”
People, Awards, and Honors
![]()
Wim Leemans of the Accelerator and Fusion Research Division was awarded the prize for “Achievement in Accelerator Physics and Technology” by the U.S. Particle Accelerator School. He was recognized for “his contributions to the development of laser wakefield accelerators, in particular the guiding of high-intensity laser beams and acceleration to high-energy of high-quality electron beams.” The prize includes a cash award of $3,000.

The American Academy of Arts and Sciences announced on April 26 the election of 213 new fellows, including Berkeley Lab physical bioscientist Hiroshi Nikaido, who studies antibiotic resistance to infectious diseases. The members are recognized as leaders in scholarship, business, the arts, and public affairs. An induction ceremony will be held in October.
The Microscopy Society of America has awarded Christian Nelson of the Materials Sciences Division its Outstanding Technologist in the Physical Sciences award. Over the last 20 years, Nelson has assisted hundreds of users at the National Center for Electron Microscopy. His services have contributed to the advancement of resolution and light-atom sensitivity. The award honors technologists from the biological and physical sciences who have made significant contributions to the field of microscopy.

Eva Nogales of the Life Sciences Division is the recipient of the second Chabot Science Award, to be presented at the Chabot Space & Science Center’s gala on June 25. The $5,000 award honors Nogales’ work in the mapping of the atomic structure of the cellular protein tubulin. By understanding the atomic structure of this protein, scientists hope to synthesize a more effective anticancer drug. Nogales is also an associate professor at UC Berkeley.

Berkeley Lab life scientist Amy Kronenberg is one of the members of a newly formed Exploration Systems Advisory Committee convened by NASA. The committee will meet quarterly to discuss such topics as technology, requirements, systems integration, and capability development.

Linda Wuy, formerly of the Earth Sciences Division and now business manager for the Chief Financial Officer, was presented by the Office of Science with an award for her contributions to coordinating the PI meetings for the Natural and Accelerated Bioremediation Research Program. The award, signed by Ray Orbach, was presented at DOE’s Office of Biological and Environmental Research annual meeting in Warrenton, Virginia.
In Memoriam
The lead electrician at the Lab’s Advanced Light Source, Robert Thomas died on April 30 after a prolonged illness. He began working at the Lab in 1990 in the Facilities Division and was later matrixed to the ALS. His spouse, Del Thomas, is also employed at the Lab in the Business Services Division.

Nahid Mahani, a specialist in waste management data validation for the Lab’s Environment, Health, and Safety Division, passed away on April 21 after a long illness. She was 50. Donations in her honor may be made to the American Cancer Society.
Berkeley Lab View
Published every two weeks by the Communications Department for the employees and retirees of Berkeley Lab.
Reid Edwards, Public Affairs Department head
Ron Kolb, Communications Department head
EDITOR
Monica Friedlander, 495-2248, msfriedlander@lbl.gov
STAFF WRITERS
Lyn Hunter, 486-4698
Dan Krotz, 486-4019
Paul Preuss, 486-6249
Lynn Yarris, 486-5375
CONTRIBUTING WRITERS
Jon Bashor, 486-5849
Allan Chen, 486-4210
David Gilbert, 925-296-5643
FLEA MARKET
486-5771, fleamarket@lbl.gov
Design
Caitlin Youngquist, 486-4020
Creative Services Office
Communications Department
MS 65, One Cyclotron Road, Berkeley CA 94720
(510) 486-5771
Fax: (510) 486-6641
Berkeley Lab is managed by the University of California for the U.S. Department of Energy.
Online Version
The full text and photographs of each edition of The View, as well as the Currents archive going back to 1994, are published online on the Berkeley Lab website under “Publications” in the A-Z Index. The site allows users to do searches of past articles.
Flea Market
- AUTOS & SUPPLIES
- ‘00 NISSAN XTERRA SE, V6, white, 81K mi, auto, 2x4, ac, CD, all pwr, ABS, running boards & roof rack, just serviced, $10,250, Steve, X6793, (925) 969-0121
- ‘96 PONTIAC SUNFIRE COUPE, 2.4 L, 5 sp, black, 88K mi, good cond, 1 owner, $2,500, Patrick, X4529, (925) 969-0176
- HOUSING
- ALAMEDA, charming, small 2 bdrm Victorian, 624 Haight Ave, avail 8/15, nr pub trans/beach/cafes, parking, basement storage, patio, w&d, eat-in kitchen, good schools, year lease, $1,500 + util, dep, Anna, 642-1206, aws@uclink.berkeley.edu
- BERKELEY, Elmwood Dist, lge sunny bdrm w/ hrdwood flrs, avail for sublet 5/22 - 8/20, $873/ per mo, neg, furn room, 1 of 2 housemates will be around for the summer, share liv/dining rm, lge eat-in kitchen, patio & stone fireplace, w&d, nr UCB/restaurants/cafes, pref female, no smok/pets, llamartin@berkeley.edu
- BERKELEY, Elmwood Dist, rm in 3-bdrm craftsman-style home, great location, walking dist to cafes/ shops/UCB, quiet neighborhood, offstr parking, nr public trans/ Rockridge BART, seeking female to share furn or unfurn room, all util incl, $650/mo, Carole, 649-8592
- BERKELEY, resid community of scientists/staff/grad students, Hearst Commons, 1146-1160 Hearst, nr UC/pub trans/BART, res parking, internet, studio townhouses w/ decks, furn can be provided at no cost, hardwd flrs, skylights, dw, ac, intercom, sec, units from $850, 848-5371, avail June-August, http://www. live-work.us/cgi-bin/artbooks/berkeley.html
- BERKELEY HILLS, small detached studio w/ sep entry, priv bath, kitchenette, no smok/pets, 1 person only, nr pub trans, offstr parking, year lease, $680/mo incl utils, Pinky, xcxc333@yahoo.com
- BERKELEY HILLS, by wk/mo, quiet furn suite, 1 bdrm/1 bth, liv rm/kitchenette, quiet, eleg & spacious, bay views, DSL, cable, walk to UCB, Pierre, 387-4015, pchew@pacbell.net
- OAKLAND, quiet Rockridge neighborhood, 2 blocks from College Ave, nr Berkeley border, furn 3 bdrm/11/2 bth, lge garden, nr BART/Lab shuttle/shopping, small park w/ play structure across str, pref visiting scholar, avail Sept 05 -Aug 06, $2,800/mo + utils, neg, Paula, 597-1233
- TARA HILLS, 2 bdrms for rent in 4 bdrm/2 bth house, share w/ a female, $550/mo, avail now, Liz, X2724, cell 685-0005, Lbear2500@aol.com
- WALNUT CREEK, 2 bdrm, fully furn, avail 5/22 - 08/22, 1 bdrm is set up as a study/office w/DSL, nr Walnut Creek BART, $900/neg + util, quiet & safe, unit is part of duplex w/ 3 other duplexes set way off from resid street, secluded, Dennis, (925) 947-5925 home, (415) 385-8356 cell, DJLamenti@lbl.gov
- MISC ITEMS FOR SALE
- WASHER, 2 year old, portable, $100, Maisha, 776-4197 cell
- WINDSURFING BOARD, $35, Sasha, X7533, (925) 937-2318
- VACATION
- PARIS, FRANCE, near Eiffel Tower, furn, eleg & sunny 2 bdrm/1 bth apt, avail by week/mo, close to stores, restaurants, trans, 848-1830
Flea Market Policy
Ads are accepted only from Berkeley Lab employees, retirees, and onsite DOE personnel. Only items of your own personal property may be offered for sale.
Submissions must include name, affiliation, extension, and home phone. Ads must be submitted in writing
(e-mail: fleamarket@lbl.gov, fax: X6641), or mailed/ delivered to Bldg. 65. Email address are included only in housing ads.
Ads run one issue only unless resubmitted, and are repeated only as space permits. The submission deadline for the May 27 issue is Thursday, May 19.