
Hydrated
Electrons Make a Big Splash
Molecular Foundry Takes
Shape
Hydrated Electrons Make a Big Splash
Science Magazine Lists Results Among Breakthroughs of 2004

Daniel Neumark
Berkeley Lab scientists appear to have settled a long-standing scientific question about water clusters — aggregates of water molecules that feature unique properties, somewhere between that of liquid water and steam. Experiments led by Daniel Neumark, director of the Chemical Sciences Division, have identified two distinct forms of negatively charged water clusters, thereby providing new insight into the fundamentally important interaction between electrons and water.
These results, along with an earlier companion paper by Neumark, were listed by Science magazine among its choices for “Breakthroughs of 2004” in its December 17 issue.
”After a century of intense scientific study, water still gives research-ers much to scratch their heads about,” the Science citation reads. “This year, a flurry of papers on the structure and chemical behavior of this familiar substance revealed results that, if they hold up, could reshape fields from chemistry to atmospheric sciences.”
Neumark is the principal author of a paper published in Science Express, the online version of Science, entitled Observation of Large Water Cluster Anions with Surface-bound Excess Electrons. The other authors are Jan Verlet, Arthur Bragg, Aster Kammrath of UC Berkeley, where Neumark is a professor of chemistry, plus Ori Cheshnovsky of the Tel Aviv University in Israel.
“We have confirmed the presence of two isomers of water cluster anions: internally solvated structures, in which a hydrated electron is localized within the cluster; and surface state structures, in which the hydrated electron is bound to the surface of the cluster,” Neumark says. “The internally solvated structures are the ones whose properties should approach those of the bulk liquid hydrated electron as the cluster size is increased.”
Hydrated electrons form when an excess of electrons are injected into liquid water. Despite being the focus of numerous studies since their discovery in 1962, there remains much to be learned about hydrated electrons. What is known is that their presence enhances the reactivity of water molecules with other molecules in a number of important chemical, physical and biological processes.
The long-held belief has been that an individual hydrated electron is confined within a small void created by a surrounding cluster of water molecules. Clusters, which may consist of as few as three or as many as 20,000 individual atoms in size, are too large to be thought of as a molecule but too small to be classified as a bulk-phases liquid or a solid. Because of their in-between size, they often make excellent subjects for learning more about the physical and chemical properties of bulk phase materials.
“In our lab, we carry out experiments that help us understand how phenomena associated with macroscopic materials manifest themselves in finite clusters,” Neumark says.
One of the key questions his group has been addressing concerns the relationship of a hydrated electron to the size of the water cluster surrounding it. How large can the cluster be for the effects of the hydrated electron to mimic the effects in bulk phase liquid, and do those effects change as the cluster grows in size? Previous studies indicated the presence of hydrated electrons in water clusters but found conflicting results as to their effect.
“The problem was those earlier studies couldn’t determine that the clusters could have either an internally solvated or a surface hydrated electron structure,” Neumark says. “Our experiment pretty much settles this issue.”
 In the traditional view, a hydrated electron (shown in red) is confined
within a small void created by a surrounding cluster of water molecules.
Berkeley Lab researchers have found an alternative structure in which
the hydrated electron may be bound to the surface of the cluster.
In the traditional view, a hydrated electron (shown in red) is confined
within a small void created by a surrounding cluster of water molecules.
Berkeley Lab researchers have found an alternative structure in which
the hydrated electron may be bound to the surface of the cluster.
Neumark and his colleagues created clusters of water anions by passing argon gas over molecules of water and heavy water at temperatures of 20 degrees Celsius, introducing the gas mixture into vacuum, and generating negatively charged clusters through the interaction of the gas mixture with low energy electrons. The clusters were then studied using a combination of femtosecond laser light and time-resolved photoelectron imaging. This application of time-resolved techniques to gas phase processes occurring on a femtosecond time scale has been one of the most important developments in chemical dynamics during the last 10 years and has yielded valuable information on the photo dissociation and reaction dynamics of molecules and clusters.
In this latest effort, Neumark and his colleagues were able to characterize an entirely new class of cluster anions with vertical binding energies (the energy required to remove an electron from its orbit) that were significantly lower than any previously recorded.
“The data are consistent with a hydrated electron structure in which the excess electron is bound to the surface of the cluster,” Neumark says. “This result implies that previously observed water cluster anions, with higher vertical binding energies, were indeed from internally solvated electrons and are therefore structurally similar to a bulk hydrated electron.”
From their findings, Neumark and his colleagues conclude that an anion water cluster needs to consist of at least 11 to 25 molecules in order to be able to have distinguishable internally solvated or surface state structures. They also found that they could create conditions that would favor the formation of one structure over the other.
Says Neumark, “By operating our ion source so that we produced colder clusters, we were able to favor the formation of surface state over the internally solvated structure. That was somewhat surprising since the internal structures tend to be more stable. For the surface structure, we’re basically attaching electrons to ice nanocrystals.”
Molecular Foundry Takes Shape

The building has come a long way since a rainy day in late January 2004 when dignitaries broke ground on the $85 million, six-story, 94,500 square-foot building, which will serve as a national user facility aimed at the development of nanotechnology — one of five new centers for nanoscale scientific research to be established at U.S. Department of Energy laboratories.
The next milestone, the welding of the steel girders, should be completed
by early February. The facility should be ready for initial user operation
before the May 2006 target date, Krupnick said. When completed, the
Molecular Foundry will house more than 200 scientists and engineers
from Berkeley Lab and around the world.

Fun Commute with Two-Wheeled Wonder

Michael Dupray on his Segway.
Michael Dupray has been called a geek and was once harassed by a group of young men on his way home from work — all because he employs a unique mode of transportation.
Called a Segway, the machine uses gyroscopic technology, enabling drivers to propel themselves forward and backward by simply leaning in that direction. Turns are maneuvered with a light twist of a grip on the handlebar.
“When I heard the kids behind me, I spun my Segway around, leaned forward and started moving towards them,” recalls Dupray, who works in the Environmental Health and Safety Division. “They were so stunned, they ran off.”
For every negative response Dupray gets, however, he receives twice as many positive comments and inquiries from passersby during his commute to and from the Pleasant Hill BART station.
And he loves to sing the praises of the Segway, which, he says, is saving him nearly $100 a month in gas. With it, Duprey can climb from downtown Berkeley to Building 75B in just 18 minutes, can drink his coffee, and, if needed, answer his cell phone, all while enjoying the smooth, silent ride of his two-wheeled wonder.
“My machine is customized with pouches, so I can also use it for trips to the grocery store or local mall,” says Dupray. “And the best part is not having to worry about parking.”
The Segway uses batteries that can be recharged in any three-pronged wall outlet. Currently, Dupray’s range is about 10 miles, but a new model of battery coming out this year will nearly double that. The 85-pound machine can reach speeds of about 12 miles per hour.
Dupray thinks the energy-efficient and environmentally friendly Segway would make the perfect augmentation to the Lab’s fleet of vehicles.
“I use it when I have to make on-site inspections,” Dupray explains. “I can put my instruments and documents in the pouches and just cruise. It only costs about 10 to 15 cents to completely charge the battery, so they’re very economical.”
While Segways are still rare at the Lab, they are growing in popularity across the country. In fact, Segway Enthusiast Groups, or SEGs, have popped up, hosting such activities as Segway polo matches.
“A lot of people buy them because they’re safe, efficient and save time,” says Dupray. “But what really seals the deal is they’re fun.”
Director Chu’s First Brown Bag on Monday

1 p.m. in Perseverance Hall. All Laboratory employees are invited to bring their lunches and their questions to the table and engage in a conversation with the Director about Lab matters. The informal get-togethers were initiated by Chu’s predecessor, Chuck Shank, two years ago in order to allow staff the opportunity to engage in a dialogue with Lab leadership.
A Quantum Look at How the Mind Works
Scientists distinguish between mind and brain
A new model of the human mind and how it works, based on the principles of quantum mechanics, offers fresh hope for victims of strokes and others who suffer from brain damage or psychological disorders. With this quantum model, neuroscience concepts and proposed therapies for neurological disorders can be tested under the same rigorous rules that are used in particle physics experiments.
“Our model provides neuroscientists and psychologists with an alternative conceptual framework for describing neural processes that works better than behaviorist therapies based upon a robotic conception of human beings,” says Henry Stapp, a theoretical physicist at Berkeley Lab and one of the three collaborators who developed this model. “Having a neurophysical model that explains how mental effort can affect brain activity should enable therapists to treat brain damaged patients more effectively.”
Stapp’s collaborators on this model are Jeffrey Schwartz, a neuropsychiatrist at UCLA, and Mario Beauregard, a psychologist with the University of Montreal.
A detailed description of the model will appear in the February 2005 edition of the Biological Sciences section of the journal, Philosophical Transactions of The Royal Society, which is published by the United Kingdom’s national academy of sciences.

Current neuroscience canon holds that the mind is what the brain does. Schwartz, an acclaimed specialist in the treatment of patients with obsessive compulsive disorder, came to a different conclusion after extensive study of positron emission tomography (PET) images of patients undergoing therapy. Contrary to mainstream thought, Schwartz concluded that the mind is a powerful and independent entity that can actually rewire the brain to serve its own needs. This rewiring ability, which he dubbed “neuroplasticity,” can open effective new pathways by which brain impairments can be healed.
“The time has come for science to confront the serious implications of the fact that directed, willful mental activity can clearly and systematically alter brain function; that the exertion of willful effort generates a physical force that has the power to change how the brain works and even its physical structure,” Schwartz stated in his groundbreaking book, The Mind and the Brain: Neuroplasticity and the Power of the Mental Force.
However, Schwartz needed a mechanism by which to explain mind–brain interactions as something other than the electrochemical activity between neurons that is set in motion by genetic dictates. This led him into a collaboration with Stapp, who for years has been applying quantum theory to understanding human consciousness.
Stapp’s investigations into the physics of human consciousness arose from his efforts as a theoretical physicist to understand the quantum features of the physical world.
“The most important innovation of quantum theory is the fact that it is formulated in terms of an interaction between the physically described world and conscious agents that are free to choose which aspect of nature they will probe,” Stapp says. “Quantum theory treats the mind (think of thoughts and sensory perceptions as our conscious agents), as an independent input that is powerfully influential with regards to the physical activities of the brain. This is a big departure from the classical conception which holds that the mind is either a passive byproduct of brain activity, or merely brain activity itself.”
It is well established that the physical activity taking place inside the brain depends heavily on chemical and ionic processes. For example, the release of a neurotransmitter, a chemical signal that connects one neuron in the brain to another (like the logic gate on a computer chip), is controlled by the motions of calcium ions. These ions are too small for their dynamics to be described through classical physics, but their activity is accurately modeled by quantum mechanics.
In the quantum model developed by Stapp and his colleagues, the brain can be viewed as an expanding cloud-like structure in a high-dimensional space with a variety of possible states, like the cloud of electrons that surround the nucleus of an atom. When the brain accepts input from the mind, it constructs a “Template for Action,” a pattern of neurological signaling activity that will cause some specific response to unfold.
Within the cloudlike brain structure that the quantum model depicts, a great many alternative Templates for Action can arise. The mind makes choices from these various possible brain states that, in strict accordance with the rules of quantum mechanics, will have actual physical effects on the neural wiring of the brain. These effects can be measured through the use of medical imaging techniques.
“With previous models of the brain, the brain completely controls the mind and any causal effects of the mind are either redundant or illusory,” Stapp says. “With our model, the mind acts on the brain, and its actions are, by virtue of basic quantum principles, not determined solely by the mechanical activity of the brain.”
As evidence that their model is an accurate representation of mind–brain interactions, Stapp and his colleagues have shown it can be used to explain numerous findings in the fields of neuroscience and psychology that cannot be explained using models of mind–brain interactions based on classical physics. For example, terms such as “feeling” “knowing” and “effort” cannot be described exclusively in terms of the brain’s material structure, but can be described in a model of the mind based on quantum principles. Having such a model should be a significant advantage to those who study and treat patients suffering from brain damage.
Says Schwartz, “Being able to show that mental effort directly affects brain mechanisms certainly enhances the context in which a therapist asks a sufferer to exert therapeutic effort. Placing self-directed neuroplasticity on a firm theoretical foundation also allows for the active aspects of human mental life to play a key role in the treatment of biological disease states.”
Adds Stapp, “Quantum mechanical rules allow choices to be made that, on the psychological side, inject new experiences, associated with a newly chosen course of action, into the stream of consciousness. On the physical side, such choices actualize brain states that contain the neural correlates of those experiences. This is the basic dynamical process that underlies the quantum approach to consciousness.”
Aerosols Overstay Their Welcome
A team of scientists has determined that carbon-containing aerosols, like an obnoxious relative who lingers a bit too long after the holidays, remain in the atmosphere much longer than previously thought, meaning they have more time to wreak havoc on Earth’s climate.
Their results, based on high-resolution images obtained at Berkeley Lab’s Advanced Light Source (ALS), will help climate scientists refine the computer models used to predict climate change. The images reveal that aerosol particles oxidize more slowly than earlier estimates indicated, which buys them more time to waft about in the atmosphere.
“These longer-lived particles will increase the carbon-containing aerosol burden on climate models by 70 percent,” says Mary Gilles, a researcher in the Chemical Sciences Division who conducted the work with Satish Myneni of the Earth Sciences Division. Lynn Russell of San Diego’s Scripps Institute of Oceanography led the research, which was published in the December 10 issue of Science.
“Our work suggests the climate models need more work, which we’ve known all along,” adds Gilles. “But this study shows us how we can improve them.”

Most people equate aerosol with hairspray and household cleaning products, but a large portion of the microscopic particles floating in the air originate from the incomplete burning of coal and oil and dust storms. Once in the atmosphere, these tiny carbon-containing particles can have a huge impact. Lighter colored organic carbon particles cool regions of the planet by scattering sunlight back into space. Other aerosol particles composed of black carbon, or soot, warm the atmosphere by absorbing sunlight and heating the surrounding air.
Impacts like these are why scientists are striving to learn how long carbon-containing aerosols remain in the atmosphere. And one way to gauge an aerosol’s ability to stay aloft is to determine its oxidation rate. Because oxidized aerosols absorb moisture and subsequently form clouds and fall as rain, the faster an aerosol particle oxidizes, the less time it spends in the atmosphere, and the less impact it has on the climate.
Hoping to learn more about these oxidation rates, the team examined four sets of carbon-containing aerosol particles that had undergone vastly different journeys. One sample of particles, collected by an airplane over the Sea of Japan, had drifted with the winds for 30 hours after originating from the factories and fields of China. Another sample, collected over the Caribbean Sea, contained aerosol particles that emanated from a dust storm that swept across Africa five days earlier. Two more samples of mostly industrial combustion emissions were collected near Princeton, New Jersey about 10 hours after the particles entered the atmosphere. One sample was collected on a foggy day, the other on a clear day.
The scientists then used scanning transmission x-ray microscopy at the ALS to analyze particles from each sample. They found that all the particles — whether they’re from an African dust storm, Asian factory, or New Jersey smokestack — oxidize at a rate of between 13 and 24 percent per day. These conversion rates are a factor of three slower than the 60 percent per day typically used in current climate models.
“The bottom line is that the rate of oxidation is slower than is currently used in models,” says Gilles. “And oxidation has a strong effect on their lifetime in the atmosphere, which has a strong effect on the global climate.”
Lab-Community Partnership Will Train Students in Energy-Efficient Technologies
Berkeley Lab is teaming with educators at Oakland’s Peralta Community College District to train students in the latest techniques of managing buildings for maximum energy efficiency. The work is funded by a three-year, $900,000 grant from the National Science Foundation.
Peralta’s Laney College has been offering a training program in Environ-mental Control Technology for three decades. Through the partnership with Berkeley Lab, a new curriculum is being developed that uses an innovative computerized simulator to give students the skills they need to commission and operate high-performance buildings with emphasis on energy efficiency and indoor environmental quality.

Rollie Otto, head of Berkeley Lab’s Center for Science and Engineering Education (CSEE), says, “The Center will support faculty-student teams from institutions across the country to participate in research that exposes them to building energy science advances and career opportunities as building operators and technicians. This will also help build leadership at community colleges for curricular improvements.”
The program currently serves about 50 new students each year. Through this new partnership, this number will increase to 600 over three years across several community colleges.
The training will utilize a new simulator tool, especially valuable in teaching students how heating, ventilation, cooling and refrigeration (HVAC&R) systems work and how to diagnose problems and find solutions.
“The are numerous gaps between the education received by building operators and technicians and current workplace needs,” says Evan Mills, a scientist in the Lab’s Environmental Energy Technologies Division. “To scope out the problem, we and our colleagues at Peralta conducted a focus group and follow-up interviews with about 40 industry stakeholders last year. They concurred that many of the problems we see today in achieving and maintaining energy savings in buildings can be traced to inadequacies in building operation and lack of awareness and knowledge about how existing systems are to be used, monitored, and maintained.”
The program targets students in two-year community college programs. CSEE also plans to support faculty and students at colleges with environmental controls technology associate degree programs, beginning with a workshop to promote the dissemination of the tools in summer 2005. The Center will also provide mentored summer research in the building sciences at Berkeley Lab for students completing associate degree programs. Finally, the team will develop and implement a standards-based physics course that provides high school students with concurrent credit at Laney College and Oakland Unified School District, in collaboration with high school teachers.
The Berkeley Lab team consists of Philip Haves, leader of the Commercial Buildings Systems Group, plus Mills and Otto. The Peralta Community College District consists of the College of Alameda, Laney College, Merritt College, and Vista Community College.
Lab’s DOE ‘Broker’ Says Goodbye
Richard “Dick” Nolan was packing up his office earlier this week, preparing to say goodbye to his 10 years as director of the Berkeley Site Office (BSO) for the Department of Energy, and to his 37 years of federal service. Now facing a busy retirement in the real estate management field, Nolan reflected on his rich career of experiences, concluding that he “couldn’t have done anything more rewarding.”
He was focused on a career in public service straight out of San Diego State University, with his bachelor’s and master’s degrees in public administration. Part-time internships during graduate school with the city and county of San Diego, a public employees’ union, and the Spacecraft Center in Houston laid the groundwork. A suggestion from a fellow intern, current Berkeley Lab procurement officer Tony Tiritilli, led him to his connection in 1968 with the Atomic Energy Commission, predecessor to the DOE.
His tenure with the AEC’s Nevada office, including a two-year intergovernmental agency exchange with the City of San Diego for environmental analysis work, lasted until 1975, when he was recruited for the Oakland Operations Office. Before he moved to Berkeley Lab to head the site office here in 1994, he served as DOE-OAK’s chief of planning, director of the Energy Research Divisions, and director of Communications. He also served Washington as deputy director of the site selection task force for the ill-fated Supercon-ducting Super Collider.
Now Dick Nolan is going home, in more ways than one. He will move to east San Diego and into the childhood home of his wife Marilyn, who was his sweetheart at Granite Hills High School. Engaged to be married in their teens, they cancelled their plans and went their separate ways. Then, in October of 2003 — a combined five marriages, three children, and 40 years later — they finally wed and are now partners in life and business. They will rely on Dick’s interest in land management, grown since he assisted in his father’s land surveying business as a kid, and Marilyn’s real estate acumen as an escrow officer, to run CAMINK Investment Manage-ment — that’s Cam, after the Nolans’ dog Camie, and Ink, short for their cat Inker.
At 60, Nolan is excited about the next chapter in his life, but no more so than his delight with the prior chapter.

View: What do you think you’ll miss most about the Berkeley Lab environment?
Nolan: The excitement of this place. There is so much energy here — a real passion for scientific discovery. Think about it. Combine the “big science” legacy of Ernest Lawrence with a first-class scientific staff, one-of-a-kind experimental facilities, the catalytic connection to the Berkeley campus, and outstanding administrative and operational support. You get an institution that rivals the intellectual curiosity and scientific discovery of any place like it in the world.
View: Can you assess the health of the Laboratory as you depart?
Nolan: The budget and human resources assets of the Laboratory are quite fit. Steve Chu will carry on the outstanding leadership tradition left by Chuck Shank. The Lab’s scientific and support staff are first rate.
I wish I could say the same for the physical plant. While there are bright spots such as the construction of the Molecular Foundry, Building 77 improvements, and the dismantling of the Bevatron, the infrastructure continues to suffer from a lack of funding. George Reyes (director of the Facilities Division) and his crew do a marvelous job of keeping things running. But the lack of funding short-changes the Lab’s ability to do first-rate science when it must be done in second-class facilities. This has been a systemic problem throughout the DOE complex during my career, and I don’t see it changing.

View: As you look back over the past decade, what particular programs and events give you the most pride?
Nolan: I am especially proud of the site office’s contribution to improvements in the Lab’s administrative and operational performance. The key has been the ability of the DOE, the Laboratory, and the University of California corporate staff to team together and agree on clear performance expectations — expectations which can be measured. The Lab has taken risks in giving us the data — good and bad — we need to assess performance. We treated that data with respect, not overreacting, and worked to find ways to improve. Sure, there have been some rough spots, but the proof is in the most recent performance results, where the Lab was judged “outstanding.”
View: The site office partnership with Lab management has been especially strong under your tenure. Cooperation and trust have never been better. What can you tell your successor to ensure that this continues in the future?

Nolan: The partnership has been extraordinarily productive. My philosophy has always been that there are ways to find solutions to problems that get in the way of getting the science done. To be successful, the BSO must see itself as a “broker” between the Lab and DOE headquarters. The DOE has a way of setting costly and sometimes inappropriate requirements that cry out for creative solutions. Since those requirements have to flow through my office, we have the leverage to find win-win solutions.
As a result, we have been successful in developing a truly effective Integrated Safety Management (ISM) program, which has led a dramatic decrease in reportable accidents; we dealt effectively with community concerns regarding the use of tritium at the Lab; we have maintained the Lab as an open Office of Science facility; we developed a “best practices” study for management benchmarks; and we constructively overhauled the Lab’s business systems. In addition, we succeeded in securing complicated off-site leases for the Joint Genome Institute and the Oakland Scientific Facility, we acquired mission-need approval for Bevatron demolition, and we launched the Molecular Foundry building project.
Using sound management principles, there’s no issue on earth that can’t be solved. In August, Steve Chu and I signed an updated set of principles rooted in three important concepts — mutual trust, respect for the roles of the DOE and the University, and high levels of communication.

View: What are the major challenges ahead for your successor, and for the Lab?
Nolan: First is competing successfully for the award of a new contract for operation of the Lab, and then adapting to a new contract and its award-term incentives. Also, the Office of Science has reorganized into a “One SC” concept, which includes significantly expanded contracting officer authority in the site office, and good working relationships will be key. The Lab will have to help the Office of Science in re-engineering its business systems, and also adapt to SC’s new lab performance appraisal process.
View: What would you like the employees at Berkeley Lab to know about the Berkeley Site Office and the value it brings to the Lab’s overall welfare and success?
Nolan: I want everyone to know how grateful I am for the 10 wonderful years I’ve spent here. I’ve enjoyed great friendships and working associations. It is my hope that you will continue to look at the BSO as your ally in finding solutions to problems and being successful. Sure, we must maintain our stewardship responsibilities and manage the contract in the best interests of the taxpayer. But we must never lose sight of the wonderful science that is driven by this place. For with all the business and operational challenges we have to tackle, none of us would be here were it not for the excitement of scientific discovery.
Berkeley Lab Slashes Injury Rate
What a difference a year makes. In FY 2004, Berkeley Lab posted the best safety record of the 10 Office of Science labs, slashing by more than 50 percent both its injury rate and the number of accidents resulting in lost workdays among its full time employees.
The success is due in large part to a labwide safety awareness campaign launched last January in response to comparatively poor FY 2003 injury rates.
“The strategies we developed have been very effective,” says Robin Wendt, deputy director for the Environment, Health and Safety (EH&S) Division, who retires at the end of January. “We focused on increasing safety in the three divisions which together accounted for more than half the recordable accidents at Berkeley Lab.”
 Safety
performance data shows dramatic improvement for Berkeley Lab.
Safety
performance data shows dramatic improvement for Berkeley Lab.
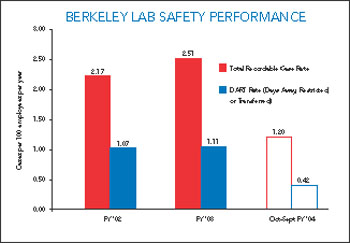
According to preliminary Office of Science data regarding FY 2004 safety performance among the 10 science labs, Berkeley Lab earned the second lowest Total Recordable Cases (TRC) rate, which is a measure of the number of injuries that require prolonged medical attention (prescription drugs, physical therapy, sutures, etc.) per 100 full time employ-ees per year. And Berkeley Lab boasted the lowest Days Away and Restricted or Transferred (DART) rate, a measure of the number of these incidents per 100 full time employees per year that require the injured employee to miss work or switch duties.
“These high rankings are only possible because our research divisions continued their very effective efforts to maintain a safe workplace,” adds Wendt.
Specifically, Berkeley Lab’s TRC rate was reduced by 53 percent in FY 2004, to 1.20 cases. In comparison, the average TRC rate among the 10 labs decreased by 15 percent during the same period. Berkeley Lab will need to reduce its rate by an additional six percent in FY 2005 to achieve its target of 1.10 cases.
Berkeley Lab’s DART rate scored even better, dropping 57 percent in FY 2004, to 0.42 cases, compared to the 22 percent drop among the 10 labs. In addition, Berkeley Lab has already reached its FY 2005 target level — one of only two Office of Science labs to be one year ahead of schedule. Improvements like these will keep Berkeley Lab focused on its mission, Wendt says.
“We are in the business of conducting science, and if the safety goal is sacrificed, everything can be lost,” says Wendt. “We want our workers to be safe, and we want our Lab to be sustainable.”
Berkeley Lab also leads the pack when it comes to radiation safety. “Of the five multi-program labs, we are the leaders in keeping radioactive dose as low as possible,” says Wendt. “We are also ahead of the Office of Science-specified pace for correcting deficiencies stemming from the January 2004 Occupational Safety and Health Administration [OSHA] audit.”
Phyllis Pei, the new EH&S division director at Berkeley Lab, is working with senior Lab management to implement new strategies to achieve even more aggressive accident reduction goals in the future.
New Acronym, Same Quality Service
TEID is out, and CSO is in.
That would be the Technical and Electronic Information Department, long-time resource for all things printed, graphic and technically written at Berkeley Lab, most of which morphed into the Creative Services Office, now under the jurisdiction of the Public Affairs Department.
Historic users of the group’s services — which include design and illustration, printing and duplication, photography, web development and design, video and production services, and technical writing and editing — need not fret that the capabilities and resources will be negatively impacted. On the contrary, the change is designed to reflect a more streamlined and responsive effort.
“We are re-evaluating the programs, with an eye toward providing our customers with the most effective service at the most reasonable price,” said Reid Edwards, head of the Public Affairs Department. “We want to make our creative services second to none, so that the products that represent this laboratory to its many audiences will be of the highest quality and uniformly reflective of our excellence.”
 Cheryl
Ventimiglia displays CSO’s new services list.
Cheryl
Ventimiglia displays CSO’s new services list.
The core of CSO will remain housed in Building 46, under the management of Cheryl Ventimiglia. A new website (http://www.lbl.gov/ LBL-PID/CSO/) describes its publications, media and professional design services.
“Our staff is excited about the change and ready to take our program to a new level of service,” said Ventimiglia. “From the smallest duplicating job to the flashiest high-end project, we will continue to provide our customers here with the most cost-efficient and attractive product, one that is price-competitive with any other agency.”
Among the many satisfied customers is Pat Dehmer, director of the Department of Energy’s Office of Basic Energy Sciences, who praised CSO last month for its work designing, laying out and printing a major report for her office. “The cover is gorgeous, and the report is compelling,” she said. “I couldn’t put it down. We will put this to good use immediately.” John Hules of Computing Sciences wrote the report.
And David Gilbert, Public Affairs Officer for the Joint Genome Institute in Walnut Creek, said, “CSO serves as one stop shopping for JGI’s communications needs. From the design that has inspired JGI’s new website, to the poster depicting DOE’s contribution to the Human Genome Project featured in a recent edition of the journal Nature, CSO’s team of talented artists, graphic designers, writers and editors keeps us looking and sounding great.”
The 12-person team offers the following services:
- Design: publications (annual reports, brochures, posters, fliers,
newsletters, websites), identity marks, production (presentations,
word processing, prepress), displays and exhibits, illustrations
(technical and 3-D) and animation.
- Photography: professional in-studio portraits and on-location
shooting, digital imaging, prints, slides, poster printing, laminating
and mounting.
- Duplicating and printing: high-speed black-and-white and color
copying, CD duplication, full-service bindery, and coordination
of full-color publication printing.
- Web development: designing, creating and updating websites, full
build-out or template development, and software training.
- Videography: videotaping of events, full-service editing and production
(effects, titles, animation, voiceover recording, sound tracks),
digital video for website or PowerPoint use, scripting assistance,
duplication services.
- Technical writing/editing: original text development, proofreading and copy editing (for journal articles, annual reports, brochures, websites, proposals, presentations, etc.), abstract submission services.
The Creative Services team includes Ventimiglia; Alice Ramirez, Flavio Robles and Caitlin Youngquist (production, illustration and design); Roy Kaltschmidt (photography and poster printing); Julie McCullough, Theresa Duque and Bruce Balfour (technical writing/editing); Faye Jobes and Mike Muller (printing and duplicating); Erik Richman (web services); Loretta Hintz (video services); and Jeanne Gerstle, CSO Administrator. CSO personnel matrixed to divisions include Greg Vierra, illustrator with the Advanced Light Source, and Jim Miller, writer/editor with Facilities.
Customers are reminded that, due to budget reductions caused by financial belt-tightening, a few services have been limited. For example, video services will only be available Monday through Thursday from 8 a.m. to 1 p.m. If needed outside these hours, arrangements should be made at least two weeks in advance. Audio-visual services to conference rooms, now provided by the Information Technologies and Services Division (ITSD), are now limited to the Building 50 and 66 auditoriums and Perseverance Hall. Services in support of the smaller conference rooms will be the responsibility of the event’s sponsoring department or division.
The Library and Archives and Records Program and the Report Coordination Office (journal publishing and programmatic scientific and technical publishing), formerly within TEID, will continue as before under the management of ITSD.
To discuss a project, request a cost estimate, or ask questions about
the services, call X6765.
Berkelely Lab View
Published every two weeks by the Communications Department for the employees and retirees of Berkeley Lab.
Reid Edwards, Public Affairs Department head
Ron Kolb, Communications Department head
EDITOR
Monica Friedlander, 495-2248, msfriedlander@lbl.gov
STAFF WRITERS
Lyn Hunter, 486-4698
Dan Krotz, 486-4019
Paul Preuss, 486-6249
Lynn Yarris, 486-5375
CONTRIBUTING WRITERS
Jon Bashor, 486-5849
Allan Chen, 486-4210
David Gilbert, 925-296-5643
FLEA MARKET
486-5771, fleamarket@lbl.gov
Design
Caitlin Youngquist, 486-4020
Creative Services Office
Communications Department
MS 65, One Cyclotron Road, Berkeley CA 94720
(510) 486-5771
Fax: (510) 486-6641
Berkeley Lab is managed by the University of California for the U.S. Department of Energy.
Online Version
The full text and photographs of each edition of The View, as well as the Currents archive going back to 1994, are published online on the Berkeley Lab website under “Publications” in the A-Z Index. The site allows users to do searches of past articles.
Flea Market
- AUTOS & SUPPLIES
- ‘99 Saturn SL2, 69K mi, ext warranty good until 04/05, auto trans, all pwr, 4 dr, Michelin tires, Blaupunkt stereo/CD, exc cond, very reliable, $6,800/bo, Deb, X7078, (415)819-1819
- ‘00 FORD MUSTANG, 2 dr coupe, red, manual 5 sp, 3.8L V6, 76.5K miles, ac, pwr drs/win, alarm, CD, new brakes, orig owner, all maint records, reg tags good through 01/06, $6,500, Julia, X6944, 981-8297
- HOUSING
- BERKELEY HILLS, bay view, furn room 17’x15’ overlooks garden, priv ent, own bthrm, quiet neighbrhd nr UC/pub trans/shops, own cooking facil in adj rm, pool table, workout mach, w/d, $850/mo, incl linens, dishes, util, phone, DSL, no smoking/pets, short stay, $300/wk, Carol, 524-6692
- NORTH BERKELEY HILLS, house w/ bay view, 4 bdrm/2 bth, hardwd flrs, liv rm, dining rm, breakfast rm, kitchen w/ appliances, w/d, enclosed sunrm off master bedrm w/multi-bridge view, water paid & gardening incl, avail now, $2,500/mo, Kenneth, 843-5253
- NORTH BERKELEY 2 bdrm/1 bth house, 1209 Henry St at Eunice, nr Gourmet Ghetto/ Solano Ave/trans, no pets/smoking, $2,200/mo, Steven, 528-2009, henryrental@aol.com, photos at http://members.aol.com/henryrental
- TARA HILLS, 2 bdrms for rent in 4 bdrm/2 bth house, share w/ a female, $550/mo, avail now, Liz, X2724, cell 685-0005, Lbear2500@aol.com
- HOUSING WANTED
- SEEKING 2 or 3 bdrm house/condo or cottage for Dutch family in the El Cerrito/Kensington/Berkeley area, April 30-May 13, visiting grandparents, B. E. Gordon, 233- 7244
- MISC ITEMS FOR SALE
- KITCHEN ISLAND, 1 drawer, 1 cabinet & two matching stools, exc cond. $150/bo, lge oval mirror, $15, Liz, X2724
- OAK DRESSER, bottom has 6 drawers + 2 smaller drawers in center compartment, top piece has lge mirror & 3 small shelves, surface in fair cond, rest in good cond, $125/bo, Christina or Ayala, 865-0863
- SOUTHWEST AIRLINES R/T voucher, exp 11/16/05, inc drink coupons, $300/ bo; 2-seat sofa, $85/bo; full-size futon w/ iron frame, $95; twin bed mattress, box & frame, bed skirt & fitted sheet incl, $95, all in good cond; floor lamp, $12, misc other items, Minmin, 847-5130
- TRUCK TRUNK TOOL BOX, aluminum tool box, low profile, fits full size Dodge pu. 64.5”x20.5”x14.5”, like new but doesn’t fit new truck, $250, Connie or Ed, (925) 413-4881, X6951
- TV, 20” Zenith, blk case, $60; Panasonic 27” TV, blk case, $85; Panasonic 25” TV, wood case, all exc pic & cond, $35; Whirlpool microwave, white, 1.2 cu-ft, exc cond, $30; Panasonic compact stereo syst w/ 3-disc CD changer, dual cass, tuner & speakers, blk, exc cond, $65, Ron, X4410, 276-8079
- YOGA MAT, sticky green-blue, exc cond, $10; solid wood chair w/oak finish from 1950s, $25, Susan, X5437, SAberg@lbl.gov
- FREE
- PHOTO SLIDE TRAYS, Sawyer/GAF type, vertical, each tray holds 100 slides, 2x2 cardboard or plastic mounts, in boxes, 58 avail, prlmic@earthlink.net
- CARPOOL
- SAN RAFAEL, space avail in long-running carpool, San Rafael/UC/LBNL, Curt, X7419
- VACATION
- TAHOE KEYS, house, 3 bdrm/2.5 bth, fenced yard, quiet sunny location, skiing nearby, great views of water & mountains, $195/night, 2 night min, Bob, (925) 376-2211
Flea Market Policy
Ads are accepted only from Berkeley Lab employees, retirees, and onsite DOE personnel. Only items of your own personal property may be offered for sale.
Submissions must include name, affiliation, extension, and home phone. Ads must be submitted in writing
(e-mail: fleamarket@ lbl.gov, fax: X6641), or mailed/ delivered to Bldg. 65. Email address are included only in housing ads.
Ads run one issue only unless resubmitted, and are repeated only as space permits. The submission deadline for the January 21 issue is Thursday, January 13.
People, Awards and Honors
Iglesia Receives Catalysis Lectureship

Iglesia
Enrique Iglesia of the Chemical Sciences Division and professor at UC Berkeley has been awarded the 2005 Robert Burwell Lectureship in Catalysis. The citation commended him for creating “fascinating stories connecting the chemistry of materials, kinetics, in situ characterization, and reaction-transport models to understand industrial catalysis and to design new catalysts … His continuous interest in industrial problems is reflected in his filing of eight patents since joining Berkeley.” The lectureship provides an honorarium and a travel stipend.
McInturff Wins Superconductivity Prize

McInturff
Alfred McInturff of the Accelerator and Fusion Research Division recently received an award from the Council on Superconductivity, a subgroup of the Institute of Electrical and Electronics Engineers. He was recognized for “continuing and significant contributions in the field of applied superconductivity.” His work focuses on conductor development and magnet design and construction and testing at various high-energy physics research institutions.
