"We came to the important realization that in the search for key players in
the
initiation and maintenance of cell differentiation, both the cell and the
microenvironment must be included," she says. Further observations
convinced her
that any alteration of the ECM, particularly the component known as the
"basement
membrane" which lies below the epithelial cells, could have serious
consequences
for cells.
"We came to the important realization that in the search for key players in
the
initiation and maintenance of cell differentiation, both the cell and the
microenvironment must be included," she says. Further observations
convinced her
that any alteration of the ECM, particularly the component known as the
"basement
membrane" which lies below the epithelial cells, could have serious
consequences
for cells.
 Between 1980 and 1981, Bissell, working with Hall, proposed a theory stating
that
the ECM was crucial for the normal functioning of cells. By implication then,
loss of or damage to the ECM could lead to malignancy. When Bissell's ECM
theory
was first proposed, there was no immediate rush to sign on to it. Conventional
scientific wisdom at that time relegated the ECM to a passive support role in
the
drama of life. The ECM was thought to serve as nothing more than an inert
scaffold upon which cells grew and developed. Now, however, a rising chorus of
scientists are echoing Bissell's belief that the ECM is a dynamic player in cell
growth and development as well as in the onset of malignancies and other
diseases. Behind this change in attitude has been a string of experiments
conducted by Bissell and a succession of collaborators over the past 15 years
that have added piece after piece to the puzzle of how ECM regulates normal
cell
function and how damaged ECM may lead to breast cancer. Though the picture is
not
complete, clear patterns have emerged.
Between 1980 and 1981, Bissell, working with Hall, proposed a theory stating
that
the ECM was crucial for the normal functioning of cells. By implication then,
loss of or damage to the ECM could lead to malignancy. When Bissell's ECM
theory
was first proposed, there was no immediate rush to sign on to it. Conventional
scientific wisdom at that time relegated the ECM to a passive support role in
the
drama of life. The ECM was thought to serve as nothing more than an inert
scaffold upon which cells grew and developed. Now, however, a rising chorus of
scientists are echoing Bissell's belief that the ECM is a dynamic player in cell
growth and development as well as in the onset of malignancies and other
diseases. Behind this change in attitude has been a string of experiments
conducted by Bissell and a succession of collaborators over the past 15 years
that have added piece after piece to the puzzle of how ECM regulates normal
cell
function and how damaged ECM may lead to breast cancer. Though the picture is
not
complete, clear patterns have emerged.
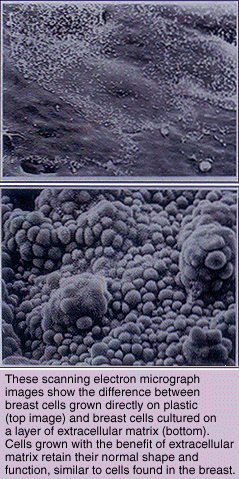
"If my hypothesis is correct, then a loss of ECM should destroy functional and morphological differentiation in cells," says Bissell. "We have demonstrated this to be the case in tissue cultures and are proving it is also true in living organisms."
One of her earlier experimental findings and one that was, in Bissell's words, "of fundamental importance," showed how the ECM, in conjunction with hormones, regulates the activities of genes within a cell's nucleus. In this case, it was how ECM induces the expression of beta-casein, an important milk protein. Bissell and her colleagues attached a bacterial "reporter" gene (one that codes for a protein marker that can be easily measured in a cell) to the part of the gene that controls the production of beta-casein. After allowing the reporter gene to become part of the breast cell's genome, they measured its activity both in the presence and absence of an ECM protein called "laminin."
Laminin interlocks with receptors on the surfaces of cells called "integrins." Like all receptors, integrins transmit chemical (and in this case possibly physical) signals across the membrane that separates the interior of a cell from the world outside. What Bissell and her colleagues found was that when laminin was present to bind to the integrin receptors, the reporter gene was 50-100 times more active than when laminin was absent. Furthermore, adding antibodies that plugged up the integrin receptors, blocked the stimulating effect of laminin.
"This clearly demonstrated that laminin binding to an integrin receptor has a direct impact on what's happening in the cell nucleus," Bissell has says. "The nucleus of the cell sensed correctly that laminin was present in the microenvironment of the cell. Perhaps the ECM tickled the receptor, which tickled the cytoskeleton, which tickled the nuclear matrix."
In more recent experiments, Bissell has explored the interactions between basement membrane ECM and genes in the cell that are associated with its destruction. In order to do this, she had to switch from working with cells grown in culture to studying transgenic mice-mice that have been genetically engineered to carry select genes from humans or other species. This work is being done in collaboration with Zena Werb, a professor of biology at UC San Francisco's Laboratory of Radiobiology and Environmental Health. The findings of two separate but related transgenic mouse studies showed Bissell's hypothesis- that loss of basement membrane ECM cripples cell differentiation and morphology-was correct not only in culture dishes, but in the body as well. The question now, she says, is how does the breakdown of ECM actively promote the development and spread of breast cancer?
Both studies involved mice that had been genetically altered to produce an abundance of stromelysin-1, one of the family of "metalloproteinase" enzymes that are known to damage basement membrane ECM. Normally activated during involution, this enzyme is an important regulator of milk production in the mammary gland. In the first study, the increased activity of stromelysin-1 destroyed the basement membrane ECM and caused the mammary glands to lose function as if the mice were already involuting. The Bissell-Werb team found that a high percentage of these mice developed tumors similar to those formed in human breast cancer, whereas no tumors were found in the normal mice.
"We destroyed the ECM inappropriately and got inappropriate results," Bissell says. "This is what we would expect if the hypothesis is correct and the ECM is crucial to a cell's ability to function properly."
Bissell and Werb suspected that the breakdown of ECM by stromelysin-1 triggered the development of genetic abnormalities in the cells. Consequently, as a followup, working with the same strain of stromelysin-1 producing mice (they also worked with tissue culture), they investigated how a breakdown of basement membrane ECM leads to "apoptosis" or controlled death of breast cells.
Controlled death is as much a part of normal cell development and functioning as controlled growth and differentiation. If a cell does not die when it is supposed to, problems occur throughout the rest of the cycle that can lead to the development and spread of cancer. In this second round of transgenic mouse experiments (as well as in the tissue culture study), the Bissell-Werb collaboration found that the destruction of basement membrane ECM accelerates the rate of apoptosis in breast cells. Absent the ECM, apoptosis in the transgenic mice started during the middle of pregnancy rather than at the onset of involution as in the normal mice.
 "A cell can either differentiate, grow, die, or become cancerous depending
on the
signal sent to it by the ECM," says Bissell. "The ECM suppresses death
by telling
the cell to grow or differentiate instead. If we remove the ECM signals, then
normal cells either die without first doing what they are supposed to, or else
they become cancerous."
"A cell can either differentiate, grow, die, or become cancerous depending
on the
signal sent to it by the ECM," says Bissell. "The ECM suppresses death
by telling
the cell to grow or differentiate instead. If we remove the ECM signals, then
normal cells either die without first doing what they are supposed to, or else
they become cancerous."
Investigating the mechanisms behind the basement membrane ECM's regulation of apoptosis, the Bissell-Werb collaboration learned that the process revolves around the expression of a protein called the interleukin-1-beta converting enzyme (ICE). When ECM was present, the activity of ICE was inhibited and apoptosis did not occur. In normal breasts, ICE increases at the end of lactation, which leads to cell death during involution.
In addition to the increased understanding about how breast cancer starts and spreads, the experiments of Bissell and her collaborators have also led to the development of a research tool that should prove valuable to future breast cancer studies. The tool is a three-dimensional assay for distinguishing normal from malignant human breast cells in culture, a distinction that in the past has been extremely difficult to make. In culture, normal human breast cells grow as well, if not better than, cells surgically removed from a malignancy, and both quickly lose their morphology and functions.
"They (healthy and cancerous human breast cells) just didn't last," Bissell explains. "You put them in culture and they clearly did not like what they saw. They did not grow very well. They did not differentiate and behave like breast cells."