|
One of the basic members of the family of cell-membrane
water channels is a protein called aquaporin 1 (AQP1). Late
in 2001, Berkeley Lab researchers announced that they had
determined the structure of AQP1 to a resolution of 2.2 angstroms,
or 22 hundred-billionths of a meter.
|
|
| |
 |
 |
|
| |
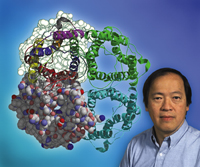
Biophysicist Bing Jap
with the water-channel protein aquaporin 1 (AQP1). Jap
led a team of researchers that determined the structure
of AQPI, revealing the means by which the protein transports
water through the cell membrane. The image shows structural
views of the four subunits of AQPI as seen from outside
the cell. |
| |
|
Such high resolution reveals the elegantly simple means by
which AQP1 can transport water through the cell membrane at
a high rate while effectively blocking everything else--even
individual protons, the nuclei of hydrogen atoms.
"Membrane proteins are a very large class of proteins;
some 30 percent of the genes in the human genome code for
them. But they are notoriously difficult to crystallize, and
only a few structures have been solved at very high resolution,"
says biophysicist Bing Jap, who led a team from Berkeley Lab's
Life Sciences Division in the difficult and painstaking crystallization
of AQP1.
The crystallized AQP1 was from bovine red blood cells, closely
similar to that in human and other cells. The team liberated
enough protein from "gallons of blood" to make 0.2-millimeter
crystals, suitable for x-ray crystallography at beamline 5.0.2
of the Advanced Light Source.
The structure of AQP1 had previously been solved to a resolution
of about 4 angstroms using electron-microscope crystallography,
which can use even smaller crystals. But at this resolution
it is impossible to see individual water molecules, and vital
features were left out or mistakenly characterized.
"AQP1 is interesting because it is so specific for water"
says Jap. "The key question was how it achieves this
specificity. Theorists had come up with lots of ideas, but
before we saw the structure in high resolution, nobody knew
how it was accomplished."
|
|
 |
|
| |
 |
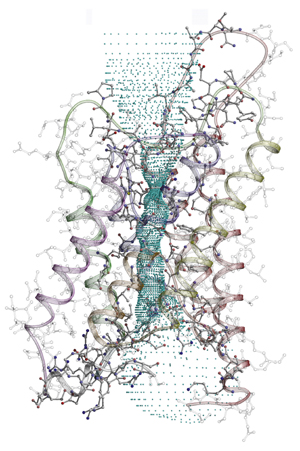
| The structure
of one of the four subunits of the water-channel protein
AQP1, embedded in the cell membrane. Each unit contains
an independent water pore (marked by blue dots). A wide
"vestibule" outside the cell (top) leads to
the pore, which is about 2.8 angstroms across at its narrowest,
just wide enough to admit water molecules. The pore widens
into another vestibule inside the cell (bottom). |
|
|
|
Architecturally, AQP1 is an assembly of four units, each
with three major structural features: each has an entrance,
or "vestibule," on the outside of the cell envelope,
connected to a similar vestibule inside the cell by a long,
narrow pore.
"The secret of AQP1's specificity is two-fold: it selects
for size and for chemical nature," Jap says. "There
is a very narrow constriction in the pore, which admits no
molecule bigger than water. To keep out molecules smaller
than water there is also a chemical filter, formed by the
specific orientation and distribution of the amino acid residues
lining the pore."
Molecules attempting to enter the channel are chemically
bound to water molecules, which are stripped away as the pore
narrows; charged species are therefore left with net electrical
charge. "The filter strongly rejects charged molecules
or ions, even as small as single protons," Jap explains.
The unique distribution of amino acid residues along the pore
wall also accounts for the channel's ability to move water
quickly, explains Peter Walian, a member of the team that
solved the structure. "It's a schizophrenic environment,
half hydrophilic and half hydrophobic"--that is, half
water-loving and half water-fearing. "Water molecules
readily get in because of the hydrophilic sites, but the hydrophobic
regions prevent them from binding too frequently."
|
| "This is
what structural biology is for -- it shows us how extremely
simple nature's solutions can be." |
|
Thus water and only water flows freely in and out of the cell
through AQP1's pores, the direction of flow depending only
on changing relative pressure inside and outside the cell.
"It's a beautiful mechanism," Walian remarks. "It's
remarkable that nobody thought of it before now."
"This is what structural biology is for," Jap says.
"It shows us how extremely simple nature's solutions
can be."
-- Paul Preuss
|



