|
|
 |
BERKELEY, CA � Researchers in Lawrence Berkeley
National Laboratory's Life Sciences Division have discovered that two
proteins, previously known for helping to construct "silent"
regions of chromosomes, also play an important but unforeseen role in
building special structures that cells need to ensure accurate chromosome
copying during cell division.
"When cells divide, they must make sure that both daughter cells
receive exactly one copy of each chromosome," says Paul Kaufman,
a staff scientist at Berkeley Lab and assistant adjunct professor of biochemistry
and molecular biology at the University of California at Berkeley. "This
process is known as chromosome segregation, and if it goes awry, cells
can lose chromosomes or acquire more than one chromosome copy." In
humans, lack of a chromosome can cause blood disorders including leukemia;
an extra chromosome 21 causes Down Syndrome.
 |
 |
| |
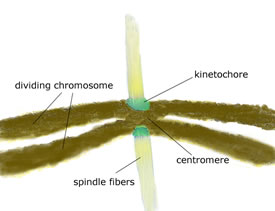
Spindles that pull the chromosome and its copy apart
attach to the centromere region: kinetochores fasten the centromeres
to the spindles.
|
As cell division begins, spindles form that will eventually pull the
original chromosomes and their copies apart into two daughter cells. These
spindles attach to constricted regions of chromosomes called centromeres:
complexes of proteins called kinetochores fasten the centromeres to the
spindles.
To elucidate this process, Kaufman and his graduate students, Judith Sharp
and Alexa Franco, focused their research on Saccharomyces cerevisiae,
the familiar single-celled organism used for centuries to ferment beer
and wine and to cause bread dough to rise. "We use yeast as a model
system in which to investigate the fundamental building blocks of chromosomes,"
Kaufman says. "The structures we're studying are evolutionarily conserved
and are much the same in many organisms, including humans."
In particular, the researchers looked at two kinds of proteins known to
be important for depositing proteins onto chromosomes. One, CAF-I (for
Chromatin Assembly Factor I), puts together nucleosomes, the fundamental
building blocks of chromosomes. Nucleosomes consist of DNA wrapped around
groups of structural proteins called histones.
Kaufman and his coworkers had previously demonstrated that CAF-I and another
set of proteins, called Hir (for histone regulatory), are important for
the formation of so-called "silenced" regions of chromosomes,
where large stretches of DNA are enveloped in protein structures that
repress gene expression. Silencing is vital to chromosome stability and
accurate segregation. In higher organisms, loss of silencing can lead
to cancer; even in yeast it can lead to developmental abnormalities and
premature aging.
"We knew that CAF-I could assemble nucleosomes in a test tube, but
it wasn't until we applied genetic tools that we discovered how much more
there was to the picture," Kaufman says. "This is the advantage
of working with yeast. It's easy to get rid of a specific gene and find
out what happens when the protein it codes for is missing."
When the researchers removed the genes that code for both CAF-I and Hir
proteins, the growth rate of the yeast slowed markedly. Moreover, yeast
lacking these genes lost chromosomes or gained extra ones hundreds of
times more often than ordinary wild yeast. Yeast that lacked only one
of the two genes was not similarly affected, however.
The delay in cell division that occurred when both genes were missing
seemed due to the activation of something called the "spindle assembly
checkpoint," a mechanism that monitors the proper attachment of chromosomes
to spindles before separation begins. This clue pointed to the involvement
of kinetochores.
Kaufman and his colleagues performed a series of tests indicating that
both CAF-I and Hir proteins are highly localized on centromeres and therefore
act directly to affect structures at these locations. Their functions
seem to overlap; thus they can partially substitute for each other if
one is missing. But when both are missing, defects in centromere structures
occur.
"This the first demonstration that proteins that control histone
deposition contribute to the formation of functional kinetochores,"
Kaufman says. "Kinetochores are essential to proper chromosome segregation
during the cell division process."
"Chromatin Assembly Factor I and Hir proteins contribute to building
functional kinetochores in S. cerevisiae," by Judith A. Sharp, Alexa
A. Franco, Mary Ann Osley, and Paul D. Kaufman, appears in the 1 January
2002 edition of Genes & Development, and is accessible online at http://www.genesdev.org/.
The Berkeley Lab is a U.S. Department of Energy national laboratory located
in Berkeley, California. It conducts unclassified scientific research
and is managed by the University of California.
|

