
Tuning
The Nanoworld
Birgeneau
Named Ninth Berkeley Chancellor
Tuning the Nanoworld
New Methods for Calculating and Controlling the Electronics of Nanostructures

Lin-Wang
Wang’s charge patching
method yields atom-by-atom electronic
maps of a nanoscale tetrapod with one
leg of cadmium selenide
and three of
cadmium telluride.
Miniature photovoltaics, quantum computers no bigger than sugar cubes, dot-sized iPods: as Berkeley Lab researchers learn to control the electronic properties of nanostructures, science fiction comes ever closer to reality.
Existing calculation methods can handle very small collections of atoms, like a molecule, or very large collections, as in bulk materials. In between — say, the few hundred to a million atoms in a nanostructure — calculations run into problems.
“One would like to use ab initio methods, which start by simply inputting a few atomic numbers and deriving the properties and behavior of the system directly,” says Lin-Wang Wang of the Computational Research Division (CRD). Alas, “anything larger than a single helium atom necessarily involves simplifications and approximations.”
For small systems physicists use a technique called local density approximation (LDA), calculating electron charge density in specific regions of a sample. But when dealing with a thousand-atom nano-structure, says Wang, “even with LDA you could spend months.”
So he and his colleagues in CRD’s Scientific Computing Group and NERSC developed a hybrid: they use LDA to determine the charge density in one small region of a crystal, then by “charge patching” extend the charge density to the entire nanosystem.
Some additional math is required, like approximating surface charge states and calculating the bonds among interior atoms. Once that’s done, LDA determines the charge “motifs” around a number of representative atoms, and the calculation is extended to the entire nanostructure via charge patching. A final step, the so-called “folded spectrum” technique developed previously by Wang, calculates the band gaps.
“The charge-patching method allows us to model thousand-atom structures with ab initio accuracy in about one hour, with the help of the Seaborg supercomputer at NERSC,” says Wang. “This gives us unprecedented power to predict the electronic and optical properties of a given nanostructure.”
Recently the new technique was applied to a novel set of branching nanostructures created in the laboratory of Paul Alivisatos of the Materials Sciences Division.
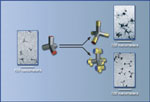
New kinds of nanostructures made by the Alivisatos group include tetrapods
of cadmium selenide (left) extended with segments of cadmium telluride (upper
right), which can be made to branch in turn (lower right).
Building branched nanostructures
“We had previously found a way to create tetrapods” — a structure with four branching “feet” or arms — “of a single semiconductor material, cadmium telluride, simply by varying the crystal phase,” says Delia Milliron, a graduate student in the Alivisatos laboratory.
Cadmium and tellurium atoms in high concentration crystallize in a cubic structure, while at low concentration the compound grows in a hexagonal structure. The two kinds of crystal are otherwise similar, with each atom bonding to four others. “Between no branching and excessive branching there’s a fine point where the tetrapods form nicely,” says Steve Hughes, another graduate student on the project.
Says Milliron, “It occurred to us that for electronic purposes we might be able to make branching structures from more than one kind of material.” The group used cadmium, selenium, tellurium, sulfur, and other constituents. “Once you have two materials, the branching possibilities increase enormously.”
A structure begins with a quantum dot, described by post-doctoral fellow Yi Cui as “a conglomeration of a few hundreds or thousands of atoms in which it’s possible to control a single electron.” The dot is made to sprout four arms of the same or a different compound.
The arms can be lengthened into nanorods containing segments of different compounds; these in turn can be induced to branch into separate rods of the same or a different compound. Electronic properties depend on the compounds involved, how they are joined, the number and arrangement of their atoms, and their proximity to other structures. “Once we had developed a rich set of structures,” says Milliron, “it was time to bring in the Computational Research Division.”
“In this case,” Wang says, “once they had synthesized the different shapes using legs and dots of different materials, we helped them figure out what they might be good for.” Nanoscale photovoltaics, quantum computers, and ultrafast transistors were among the possibilities. “In the future, by modeling a proposed system in advance, we could help decide what’s possible and how to control their results.”
“Colloidal nanocrystal hetero-structures with linear and branched topology,” by Delia Milliron, Steven Hughes, Yi Cui, Liberato Manna, Jingbo Li, Lin-Wang Wang, and Paul Alivisatos, appeared in the July 8, 2004 issue of Nature.
Birgeneau Named Ninth Berkeley Chancellor

The University of California Regents have named University of Toronto president Robert J. Birgeneau to be the ninth UC Berkeley chancellor, replacing Robert Berdahl, who is retiring. Birgeneau, 62, a native Canadian who earned his Ph.D. in physics from Yale University, is a former dean of science at the Massachusetts Institute of Technology, where he still maintains a research laboratory. He is internationally renowned for his prize-winning studies in solid state physics, and is an outspoken advocate for diversity and equity in the academic community.
“I genuinely believe that the University of California at Berkeley is simply the very best public teaching and research university in the world, and my responsibility as chancellor will be to ensure that that remains true,” Birgeneau said in a press conference following the announcement of his appointment by UC President Robert Dynes.
Dynes, who worked with Birgeneau at AT&T Bell Laboratories in New Jersey and has remained a personal friend, was effusive in praising his fellow Canadian physicist.
“Everything Bob Birgeneau has done has prepared him to be chancellor of UC Berkeley,” Dynes said. “He is a distinguished scientist of the highest academic caliber, known internationally as a leader in his field. He is a compassionate and courageous man who possesses a deep commitment to social equity and to the social responsibilities of a public university. He also is one of the mostly highly sought-after leaders in higher education today, and we are extremely proud to be bringing him to the University of California.”
New Berkeley Lab Director and fellow Bell Labs alum, Steven Chu, was also effusive in his praise of Birgeneau.

Robert J. Birgeneau (center) talks to reporters after his appointment
as UC Berkeley chancellor. Flanking him are UC President Robert Dynes (left)
and outgoing Berkeley Chancellor Robert M. Berdahl.
“I am delighted that Bob Birgeneau is becoming the new chancellor of UC Berkeley,” Chu said. “I have known Bob for over two decades as a colleague and friend. He is a terrific scientist and academic leader, and I am thrilled that I will be working with him in the coming years.”
Birgeneau received his bachelor’s degree in mathematics from the University of Toronto in 1963 and his Ph.D. from Yale in 1966. He served on the Yale faculty for one year, then spent a year at Oxford University through the National Research Council of Canada. He was a member of the technical staff at Bell Laboratories in New Jersey from 1968 to 1975, before joining MIT as a professor of physics. He was named head of the physics department in 1988, and in 1991 he was appointed dean of science. He became the fourteenth president of the University of Toronto on July 1, 2000.
Birgeneau is perhaps best known at Berkeley Lab for a Department of Energy-commissioned panel he chaired in 1997 that reviewed DOE’s four major synchrotron radiation facilities, including the Advanced Light Source. Criticisms in the eponymous report galvanized this Laboratory to take corrective actions that led to significant improvements in the relationship between the ALS and its users, and a glowing evaluation in a follow-up DOE review three years later.
Former Berkeley Lab Director Charles Shank, who led the response to the Birgeneau report, had nothing but good words for the new chancellor-designate.
“Bob Birgeneau is an outstanding educator, leader and scientist,” Shank said. “UC Berkeley is very fortunate to have a person of this stellar quality come to lead the campus at this challenging time.”
Birgeneau is expected to take office around Oct. 1. In addition to serving as chancellor, he will hold a faculty appointment in UC Berkeley’s department of physics. Birgeneau’s research has focused on understanding the fundamental properties of materials, beginning at the level of a single atom and building up to macroscopic dimensions. For this work, he has received numerous honors, including the Oliver E. Buckley Prize for Condensed Matter Physics and the J.E. Lilienfeld Prize of the American Physical Society. He is a fellow of the Royal Society of London and the Royal Society of Canada.
Joining Birgeneau in Berkeley will be his wife Mary Catherine. They have four grown children. “I am eager to begin working with everyone in the Berkeley community — faculty, staff, students, alumni, and the broader community,” Birgeneau said. “Together, we will continue this university’s world-renowned tradition of producing cutting-edge research, creating new knowledge, and teaching and developing tomorrow’s leaders.”
Director Chu’s First Week on the Job

Lab Director Steve Chu took some time during his
first few very busy days on the Hill for an informal photo session
in his new office. He took over as Berkeley Lab's sixth director on Monday,
by which time he had already introduced himself to Lab employees
via a short video (see Monday’s “Today at Berkeley Lab.”)
Passionate About that Taco Salad

Tuesday is a very special day here at the Lab. It’s when the cafeteria serves up its most popular dish: the Fiesta Taco Salad. At noon each Tuesday, a line of employees starts forming, wending its way past the salad and sandwich bar. The faithful patiently wait their turn, craving the delightful mélange of lettuce, ground turkey, taco chips and other mouth-watering toppings, all smothered in a creamy dressing.
But the troops were caught off-guard a few weeks ago when some of the ingredients in the taco salad were changed. Because of the rising costs for kidney beans and olives, cafeteria manager Jerry Roselius replaced them with garbanzo beans and corn in order to maintain the price of the salad. Little did he know his actions would set off a near panic.
Among those shocked by the substitutions was Mark Sippola, with the Environmental Energy Technologies Division. “A few coworkers and I have an admittedly absurd devotion to the taco salad — it’s our Tuesday tradition,” he says. “When the recipe changed, we were disappointed.“
Assuming others at the Lab were feeling just as strongly, he decided to start a petition. He set up camp in the cafeteria lobby and garnered more than 156 signatures.
At the same time, other cafeteria customers were providing Roselius with verbal feedback about the changes. “It’s always a delicate balance,” he explained, “between giving customers what they want and keeping an eye on our business needs. But I took what the employees had to say to heart.”
Although Roselius said he never actually received Sippola’s petition, he knew something had to be done to keep the Lab’s diehard taco salad fans happy. So, a compromise was struck: he would restore the original ingredients, but increase the price by 25 cents. In addition, employees who request “extra” ingredients will now be charged an additional fee ($1 for meat and 50 cents for toppings).
“I’m pleased with the outcome,” said Sippola. “It’s great to have the original recipe back, and the extra 25 cents is a small price to pay.”This Month in Lab History
Back to the Future: 1964
Super duper printers — beat that!
Forty years ago, in August of 1964, the Lab’s magazine the Magnet announced the introduction of “the world’s fastest printing machine.” The electrosensitive printer, introduced at Livermore Labs, printed one line at a time from magnetic tapes carried on what was then-standard IMB tape transportation. A paper transport moved the paper from a supply holder past a printing head and on to a folding device. The key was a then-revolutionary, dry-process electrosensitive paper that was smudge resistant. Sounds a little primitive? Think again.
The machine, which cost a mindboggling $350,000 (that’s 1964 dollars!) could print at a lightning-fast speed of 60,000 characters a second, the Magnet wrote, or about eight letter-sized pages a second. By comparison, today’s top-of-the line printers pride themselves with a whopping 22 or 24 pages a minute. That’s more than 16 times slower than the “outdated” technologies of the early 1960s. Now if only we could afford to go down memory lane!
Meanwhile, in the computing world …
The same issue of the Magnet announced yet another technological milestone. The computing capability of Berkeley Lab was about to double during the summer of 1964 following the installation of a new IBM 7094 computer and the conversion of an older model to a 7094-II. Housed in Building 50, the machines ran on 12 tape drives for input and output operations. Most importantly, they featured a “diprogramming” system, dubbed STEREO, which enabled the 32-bit computers to switch back and forth between two programs held in memory, allowing the computers to work on — gasp — two separate problems at the same time.
Today’s state-of-the-art IBM supercomputers at NERSC pride themselves on 6,656-processors and projected peak speeds of 10 teraflops, or 10 trillion operations per second.
“Since the mid-sixties, computing technology has grown by a factor of 100 every decade,” says David Bailey, chief technologist for NERSC. “That’s a factor of 100 million in computing power for a single computer. Back in 1964 they strung together two computers. Now in effect we string together 6,000 computers.”
For those of us too computationally-challenged to envision all that teraflop power, just consider how many applications you have open simultaneously on your modest desktop on an average workday.
And shuttle buses were on their way … Out?
August of 1964 was an eventful month. Not only did technology advance by leaps and bounds, but the Lab’s Plant Services Department decided to do away with that outdated shuttle bus system, and replace the clunky buses with a radio-dispatched taxi service. The reason for the upgrade, the Magnet reported, was that shuttle traffic had decreased due to “the convenient new parking lots,” making shuttle buses obsolete. Should employees need a ride, however, not to fear. The bus drivers would be on hand to drive two station wagons up and down the Hill while saving the Lab $23 a day.
“Thus, most bus-riding employees will be able to stay on the peak-hour vehicles all the way to the office door, instead of transferring to a shuttle as they do now,” the Magnet announced.
Taxi, anyone?
The View Takes Short Summer Break
Every summer, the staff of the View sets aside some time to regroup, plan for the next fiscal year, and consider better ways to serve our readers. As a result, the paper will take a brief hiatus, skipping the second issue in August. We’re all here, however, preparing the next issue of the View, which will come out on Sept. 3, maintaining a regular publication schedule thereafter.
New EH&S Director ‘Passionate’ About Lab

Phyllis Pei (right), the new head of EH&S, is pictured here during
the announcement of her appointment alongside former Director Charles Shank,
Deputy Director for Operations Sally Benson, and Deputy Director Pier Odonne.
On August 16 Phyllis Pei will become the next director of Berkeley Lab’s Environment, Health and Safety (EH&S) Division. Her appointment was announced to EH&S staff on July 22 by former Lab Director Charles Shank.
“We at Berkeley Lab look forward to welcoming Ms. Pei next month,” Shank said. “Safety will surely be on the mind of the new Lab Director, and today I am very pleased to announce a truly qualified and important leader for EH&S.
“At the same time, I want to acknowledge the important contributions of Robin Wendt as acting division director and express to him my personal appreciation for his dedi-cated leadership of the EH&S Division over the past several months,” said Shank. “All of us are in his debt.”
Pei’s appointment follows a nationwide search for a successor to David McGraw, who has been division director for Business Services since October. Pei has extensive leadership experience in environment, health and safety programs in private industry, consortia and national laboratories, both in the U.S. and overseas. She is currently serving as EH&S senior director at Applied Materials, a semiconductor company in Santa Clara, where she manages the environment safety and health of 12,000 employees and more than 200 worldwide facilities containing R&D, manufacturing, training, field service, refurbishing and sales operations. She is also responsible for product safety for more than 18,000 pieces of semiconductor manufacturing equipment installed at customer sites.
In introducing herself to the audience, she listed several reasons why she is excited to assume leadership of the Lab’s EH&S Division.
“First, I’m very passionate about the mission of the Lab,” said Pei. “Secondly, in the past few months of interviews, it became very clear to me that everyone is highly committed to safety. It’s very clear that EH&S has a seat at the table. And finally, I view this job as a personal challenge. I want to bring my past industry and consortium experience to the nonprofit sector.”
This past experience also includes working at International SEMATECH, where she was a key leader in developing the ES&H Technology Roadmap for Semiconductors. And as project manager at Sandia National Laboratories in Albuquerque, she developed and implemented environmental programs and prepared for the DOE Tiger Team audits.
Pei received a B.A. degree in chemistry from the University of Dallas in 1975, an M.A. degree in biochemistry from the University of Texas Health Science Center in 1977, and an M.B.A. from the University of Dallas in 1980.
Strawbale Building Inspires Visitors at Home and Abroad

Visitors and dignitaries gather outside the Strawbale Building during the
grand opening. Photo by Roy Kaltschmidt, TEID
A showcase for ecologically-sustainable building techniques, the long-awaited Strawbale Building — a 1,300 square-foot addition to the Shorebird Park Nature Center on the Berkeley Marina — has attracted more than 50 visitors a day, many of them children, in the month and a half since it opened its doors to the public. The project to build this innovative structure began in 1996 as a collaboration involving the City of Berkeley, Berkeley Lab, and the Department of Energy, among others.
“Where better than in Berkeley to have a ‘green’ building?” said Mark Seleznow, the city’s parks director, at the grand opening on June 19. “The city took a risk here — to innovate.”
The new building, peacefully situated among trees near the water’s edge, houses a visitors’ center, an outdoor classroom, and additional office space for the Nature Center. The structure supplements a City of Berkeley-owned portable building that is used for environmental education.
Its primary building material is rice straw bales, a waste byproduct of the agricultural industry. Also used in the spirit of environmentally-conscious construction were passive solar design, building-integrated photovoltaics, and hot water solar panels. Various recycled materials were used for foundations, framing, roofing, and doors.
“We are finally working in the new office,” says Patty Donald, program coordinator for Berkeley’s Marina Experience Program. “The beautiful shape of the building alone draws people in. Since the project started two years ago we’ve had over 2,000 visitors.”
Among them, Donald says, was a delegation of 35 people from China’s housing industry association, who toured the facility and attended a lecture in the hope of implementing this technology back home.
The education project is run by the Marina Experience Program, which will teach children about the ecology of the region. The program plans to incorporate concepts from the building’s construction into its educational programs.
“We offer information, including a photo album and a materials binder, and show examples that allows everyone to make a choice to take care of the Earth,” Donald says.
Construction of the building started in 2002, five years after Berkeley Lab presented the city with a check for $25,000 towards the project. In addition to DOE money, funding came from diverse sources, including corporate and private donations, local and state government grants, private foundations, and the City of Berkeley. Van Mechelen Architects and Dan Smith and Associates led the design team for the building.
A New Advance In Gallium Nitride Nanowires

Peidong Yang of Materials Sciences is the lead researcher for the project.
A significant breakthrough in the development of the highly prized semiconductor gallium nitride as a building block for nanotechnology has been achieved by a team of Berkeley Lab and UC Berkeley scientists. For the first time, researchers were able to control the direction in which a gallium nitride nanowire grows. Growth direction is critical to determining the wire’s electrical and thermal conductivity and other important properties.
“Our results will come as a surprise to those who have said that growth direction can’t be controlled, that you get what you get when you grow semiconductor nanowires,” says Peidong Yang, a chemist with the Materials Sciences Division and a professor with UC Berkeley’s chemistry department, who led this research.
Nanotechnologists are eager to tap into the enormous potential of gallium nitride for use in high power, high performance optoelectronic devices. Already, single-crystalline gallium nitride nanowires and nanotubes have shown promise in blue light-emitting diodes, short-wavelength ultraviolet nanolasers, and nanofluidic biochemical sensors.
Yang and his research group have been pioneers in the fabrication of semiconductor wires that measure only a few nanometers in diameter but stretch out to several microns in length. For this latest work, they grew single-crystal gallium nitride nanowires using a metal–organic chemical vapor deposition (MOCVD) technique that was similar to an earlier technique they used to produce nanowire lasers. Previously, Yang and his group demonstrated the ability to control the size, aspect ratio, position, and composition of their nanowires. Now they’ve added the ability to control crystallographic growth direction through the selection of a choice substrate.
Explains Yang, “In nanowires made from the exact same gallium nitride material but grown on different substrates, light emission was blue-shifted by 100 meV (milli-electron volts).”
For this study, Yang and his group used substrates of lithium aluminum oxide and magnesium oxide. The crystals of both materials are geometrically compatible with gallium nitride crystals. However, whereas the lithium aluminum oxide features a two-fold symmetry that matches the symmetry along one plane of the gallium nitride crystals, the magnesium oxide has a three-fold symmetry that matches gallium nitride symmetry along a different plane.
As a result, when a vapor of gallium nitride condenses on either of these substrates, the resulting nanowires grow perpendicular to the substrate, but aligned in a direction unique to each substrate. Because of the different growth direction, cross sections of the gallium nitride nanowires grown on lithium aluminum oxide form an isosceles triangle, while the cross sections of those grown on magnesium oxide are hexagonal.
“Our goal is to put together a generic scheme for controlling the directional growth of all semiconductor nanowires,” says Yang. “When we can do this, we will be able to answer some important fundamental questions and start making real devices.”
Yang believes that he and his group are within a few months of being able to produce a light-
emitting diode, a transistor, or a hybrid nanowire-thin film laser.
A report discussing these research results has been published in the online edition of the journal Nature Materials. In addition to Yang, co-authors of “Crystallographic alignment of high-density gallium nitride nanowire arrays” are Yanfeng Zhang, Donald Sirbuly and Jonathan Denlinger of Berkeley Lab, and Tevye Kuykendall, Peter Pauzauskie and Joshua Goldberger of UC Berkeley.
A Push-Pull Approach to Proteins
Researchers learn the biophysical properties of bacterial condensing
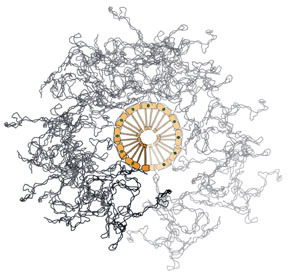
Model of the DNA condensation mechanism: MukBEF complexes are depicted
in orange, DNA as a thin black line, and the two ATPs between adjacent
heads as a small green circle. (Image reprinted with permission
by Science magazine)
By stretching a poorly understood protein like a rubber band, a team of Berkeley Lab and UC Berkeley scientists is learning how the protein and its cousins perform some of life’s most fundamental tasks. Their work — the first look at the biophysical properties of a condensin — appeared in the July 9 issue of Science magazine.
Condensins help in the final stages of folding coils of DNA into chromosomes. They are found in all organisms, from bacteria to people, and are part of a family of proteins responsible for other critical jobs such as DNA repair and meiosis. Despite its importance, condensin was discovered only 10 years ago and scientists are only now beginning to understand how it works.
“If we can understand how condensin works, it will help us understand how a large group of very important proteins work,” says Carlos Bustamante, a biophysicist with the Physical Biosciences Division and UC Berkeley’s department of molecular and cell biology, who is also a Howard Hughes Medical Institute investigator. “But we couldn’t study the protein in a bulk assay as is normally done, because such an assay doesn’t exist for condensin. So we took a risk.”
Instead, they analyzed its structure by stretching a single molecule of DNA, which had been condensed into a fiber by the binding of many condensin molecules, until some of the bonds within the condensin molecules snapped. This effect, like pulling a series of slipknots out of a rope, enabled the team to measure the strength of the broken bonds and infer how the condensin molecules are pieced together in the fiber.
“We were able to study, very clearly, how each one of the condensin molecules bound to the DNA,” says Bustamante. “And this gives us hope that we can decipher how a large family of proteins interacts with DNA.”
Bustamante conducted the research with Nicholas Cozzarelli of UC Berkeley’s department of molecular and cell biology and Steve Smith of the Howard Hughes Medical Institute. Bustamante is also part of an interdisciplinary Physical Biosciences Division initiative called Microscopies of Molecular Machines (M3), which uses conventional and newly developed microscopy techniques to probe the inner workings of molecular machines.
Earlier protein crystallography and electron microscopy research had revealed that a single condensin is about 100 nanometers long (one nanometer is one-billionth of a meter) and shaped like a nutcracker, with two rods ending at a globular domain or head, joined by a central hinge. In addition, genetic and biochemical experiments had revealed that the proteins are closely involved in maintaining the compact organization of DNA inside cells.
Although this work went a long way in describing condensin’s biochemical makeup, scientists didn’t understand the protein’s mechanical properties, or how it helps engineer one of nature’s greatest packing jobs. In DNA folding, DNA is rolled into nucleosomes, nucleosomes are coiled into 30-nanometer wide fibers, and these fibers are attached to condensins, which oversee the final compacting and folding necessary to squeeze the fibers into chromosomes. This last step puts the finishing touches on a process that somehow compresses about one meter of DNA into every human cell.
To find out how condensin helps facilitate this feat, the research team allowed the Escherichia coli condensin, called MukBEF, to bind to a single DNA molecule strung between two microscopic beads. In the presence of ATP, the condensin molecules greatly reduced the dimensions of the DNA molecule by condensing it in an ATP-dependent manner. Next, the researchers slowly stretched this condensed fiber, gradually subjecting the bonds holding the proteins together to greater and greater stress. At 17 piconewtons (one newton is approximately the weight of one apple, and one piconewton is the weight of one red blood cell), they observed a rapid-fire series of ruptures as the intramolecular contacts between the adjacent heads of a condensin molecule broke apart. At the same time, the intermolecular interactions between the heads of adjacent condensin molecules on the DNA, and their bonds to the DNA, remained intact.
In addition, when the researchers reduced the stress on the fiber by moving the two beads closer together, the protein accordioned back to its original shape, even if the excess free ATP in solution had been removed. In other words, after being deformed microns in length, the filament returns to its compact structure without the addition of (ATP) energy.
“We were able to directly probe the forces that maintain the structure of the protein assembly on the DNA,” says Bustamante. “And from this mechanistic analysis, we could infer the way in which the protein assembles itself into a larger complex.”
Based on these results, Bustamante, Cozzarelli, and Smith conclude that MukBEF forms a regularly ordered filament linear DNA, in which low-energy interactions are broken and high-energy attachments survive. These properties may also help explain how close relatives of condensin maintain the genome through jobs such as homologous DNA repair, in which broken DNA ends are repaired using information from intact DNA. Perhaps the contortionist-like protein pulls the broken and unbroken DNA strands closer together so they can exchange information. Although more work needs to be conducted on this family of proteins, such as measuring the precise energy involved in the molecular interactions holding them together, this initial inquiry into a condensin’s biophysical traits sheds light on the inner workings of life.
Science Careers Start with a Dream — and an Internship

High school student Derek Francis moved a step closer to his planned career
in bioscience while working on cancer research at Berkeley Lab this summer.
They peered into microscopes, built lasers, collected and entered data, taught girl scouts how to build robots, and helped scientists all over the Hill with projects ranging from cancer research to astrophysics. For most of these young people interning at Berkeley Lab this summer, the experience of working alongside world-class scientists is a stepping stone to future scientific careers. Some came here with their sights set on a given field of study, while others still seek to discover their own path. Some come from local high schools and colleges, others from across the country. Some have never tried their hand at science outside the classroom, while others return year after year. Whatever their background, what they all have in common is the excitement of learning and working in a pure research environment.
“I plan to major in biochemistry, so it was great to be able to have real-life laboratory experience,” said Derek Francis, a junior at Arrowsmith Academy in Berkeley who worked in Mary Helen Barcellos-Hoff’s laboratory on breast cancer research. “Here you get hands-on experience, work with real chemicals and lab equipment, such as a protein blot scanner.”
This summer Berkeley Lab hosted a record number of students — 143 of them — participating in variety of internship programs, most of them sponsored through the Lab’s Center for Science and Engineering Education (CSEE). This was the biggest high school contingent in five years (43 students from 21 different schools) and the largest number of undergraduates ever — that in spite of the fact that the DOE could only provide financial support for a fraction of them.
 Lab
astrophysicist George Smoot demonstrates a wall chart of the history of
the universe to teacher Laurie Kerrigan of Mercy High School in San Francisco,
who participated in the Pre-Service Teacher program this summer. Kerrigan
teaches physics as well as a wide range of other math and science classes
to students in grades 9-12.
Lab
astrophysicist George Smoot demonstrates a wall chart of the history of
the universe to teacher Laurie Kerrigan of Mercy High School in San Francisco,
who participated in the Pre-Service Teacher program this summer. Kerrigan
teaches physics as well as a wide range of other math and science classes
to students in grades 9-12.
“We had an increase in the number of applicants — some incredibly talented young people — and we tried to place as many of them as possible,” said Rollie Otto, head of CSEE. “What made this possible is the increase in division groups willing to provide support during the summer through programmatic funding. The researchers recognize the quality of these students and their value to their own programs. Now more investigators are looking at the rich pool of applicants online, not waiting for a call from CSEE.”
Students are teamed with mentors and pursue projects on their own, with partners, or in small groups, learning the value of teamwork and collaboration. “Before Minx came into my life I only had solo projects,” laughed high schooler Lamont Harper during his final presentation on July 30, referring to his project partner Minx Manuel. “Together we updated trauma kits and replaced fire extinguishers all over the Lab.”
CSEE-sponsored intern programs include the High School Student Research Participation Program; the Science Undergraduate Laboratory Internships (formerly the ERLUF program) and the Community College Initiative, both funded by the Department of Energy; the Faculty and Student Team (FaST) Program, which is supported by the DOE in partnership with the National Science Foundation; and DOE’s Pre-Service Teacher (PST) program, which provides educational training and research experience to undergraduate students interested in future math and science careers.
Among those who attended the final presentation of the high school interns last Friday was Assemblywoman Loni Hancock, who represents cities in Alameda and Contra Costa Counties. A strong advocate for improving educational opportunities, she served as one of President Clinton’s top education officials from 1994 to 2001, when she headed the Western Regional Office of the U.S. Department of Education. Her presence at the concluding event of the high school internship program at Berkeley Lab reinforced the value of such educational initiatives to the future of science education.
The rich educational opportunities available at the Lab are extended not just to students, however. Teachers get in on the act through the Lab’s Science Teacher Professional Development Program, which allows educators to update their own science and technology knowledge and transfer it to the classroom. In turn, their experience may spark the interest in science among other young people, giving birth to yet another dream — and perhaps someday a future internship or career right here at Berkeley Lab.
Computing Sciences hosts students from around the world

Left to right are: Ryan McKenzie of the University of Kentucky, Merten
Leupolt of the University of Technology, Germany, Maria Robinson of the
University of Toronto, Canada, Lisa Cowan of Mills College in Oakland, and
Konrad Malkowski of Pennsylvania State University.
In addition to the intern programs run by CSEE, some divisions host students on their own. The largest such group is sponsored by the Computing Sciences Summer Student Program, which gives students an opportunity to gain relevant research experience while pursuing their degree. This year Computing Sciences is hosting 16 students from various countries and universities in the United States for 12 weeks during the summer. The interns partner with one or more staff members on research projects, many of which they develop during the program and will then use as the basis for their theses.
“I’ve learned a lot this summer,” said Ryan McKenzie who is working closely with Tony Drummond and Osni Marques in the Computational Research Division. “And I’ll take what I’ve learned with me when I return to the University of Kentucky.”
Undergraduate Poster Session
Undergraduate students conducting research at Berkeley Lab this summer will present their results during a poster session to be held in the cafeteria on Tuesday, Aug. 10 at 3 p.m. More than 90 posters will be on display, and the event is expected to draw attendance from more than 200 Berkeley Lab scientists.
Flea Market
- AUTOS & SUPPLIES
- ‘00 MAZDA MIATA, silver, auto, ac, ABS, hardtop, new tires, orig owner, exc cond, 44K mi, $14,000/bo, Ka-Ngo Leung, X7918, 245-7517
- ‘99 FORD TAURUS, great cond, very clean, 89K mi, pwr win/doors, ac, cass stereo, $4,850/bo, David, (925) 625-5588
- ‘90 BMW 735i, 112K mi, silver, auto, leather, sunrf, ac, pwr win & seat, exc cond $5,900, Eran, (925) 957-1918, (925) 212-7394 cell
- ‘90 CHEVY BERETTA, outstanding cond, good tires, much new work, $1,850, Robin, (925) 838-6956
- ‘90 TOYOTA COROLLA DX wagon, 164K mi, blue metal, 5 sp man, ac, 35 mpg, good cond, $2,100, Akos, X6706, 841-4518
- WHEELS & TIRES, 4 alum slotted mag wheels w/ like-new Nankook 9.50-16.5 tires, wheels are 9.75x16.5 w/ 8 lugs on a 6.50 bolt circle, incl new 9/16” lugs & locks, Troy, X7927, $200/bo
- HOUSING FOR RENT
- ALBANY, 2/2 condo, fully furn, spacious 1,234 sq ft, sunny, quiet & eleg, sleeps 4, laundry rm, views, pool, exer & rec rms, 2-car garage, nr El Cerrito Pl/BART/public trans, 848-1830, gfchew@mindspring.com
- ALBANY, unfurn sunny newly renov 3 bdrm/ 1 bth upper unit of a dup, nr Solano Ave, shopping/El Cerrito Pl & BART, schools, avail 8/16, beautiful, http://www.realtyadvocates.com/625adams.html, $1,950/mo +dep+last mo rent, no pets, Bryan/Valerie, 985-0634
- BERKELEY, 1 bdrm furn apt nr UCB, sunny, quiet, safe, Elmwood resid area (nr Ashby/College), walk to UC/pub trans, split-level, hill view from lge terrace, fully furn include linen, dishes, hi-fi, VCR/DVD, microwave, pref one responsible, mature, nonsmoker vis scholar, avail last week of 8/04 until 9/05, dates flex, $1,050/ mo include garage, Agee, agblako@att.net
- BERKELEY, beautiful, very large studio apt in historic Maybeck bldg close to the Claremont Hotel, view of city, quiet neighborhood, plenty of parking, all kitchen amenities inc dw, built-in cherry wood bed, bureau, desk files, etc, $1,100/mo incl util, viewing from 8/1-8/6 and after 8/13, avail immediately, Carol, 548-7818, c.kusmierski@sbcglobal.net
- BERKELEY, house avail 9/1/04 through summer ‘05, off Grizzly Pk, 12 min from LBNL, 3 bdrms, house recently remodeled, beautiful rural area, reasonable rent, John, spence@asu.edu
- BERKELEY, lge 3 bdrm fully furn house, nr UC/public trans/BART, 1-yr lease, $2,000/mo+half util, 1st+sec dep of $3,500 (fully refundable), Eliene, 684-8279
- BERKELEY, newly remodeled studio apt, downstairs in home, avail 9/1, nr LBNL, Lawrence Hall/ pub trans, priv entr, bay view, garden, kitchen, all utils, cable TV, internet, $1,000/mo+occasional pet/garden sitting, John, JLMcCarthy@LBL.gov, X5307, 841-7875
- BERKELEY, Elmwood dist, master bdrm in 4 bdrm/3 bth, 2-story house, starting 9/1 neg, 25x12.5 incl priv bthrm, 2 closets, double bed, dresser, central heating, laundry, dw, full kitchen, DSL, linens, nr rest/ shops/campus/BART/shuttle stop, wooden flrs, basement, easy parking, sm backyrd, share w/ 3 grad students, 9-mo lease, Sophie, $800, sdumont@berkeley.edu, 849-2444
- BERKELEY HILLS, beautiful 1 bdrm/ 1 bth, fully furn apt, avail 8/1, spacious liv rm w/ din area, cable TV, w-w carpet, marble bthrm, modern, fully-equip kitchen, priv patio, built-in woodwork, recently remodeled, $1,090/mo+$50 util, nr UC, non-smok, 524-8308, Ivankash@hotmail.com.
- KENSINGTON, fully furn 3 bdrm home, view, quiet setting, 1 cat, avail for vis scientist during fall term, 8/1 flex, $1,600/mo +sec dep, Ruth, 526-6730
- NORTH BERKELEY, by wk/mo, fully furn 1/1 flat, quiet, spacious, dish TV, laundry rm, priv garden, gated carport, walk to LBL shuttle/UCB/pub trans, BART, downtown, 848-1830, gfchew@mindspring.com
- OAKLAND, Glenview dist, triplex, 2 bdrm apt to sublet as a 1 bdrm apt, flex move in/out dates, $550/mo & $900 when you have the apt to yourself, fully furn, dep neg, nr rest/public trans, non-smok, bukhvalo@haas.berkeley.edu, Barbara, 530-1866
- POINT RICHMOND, nr shops/rest/ cafes/theater, nice inner courtyrd w/lush garden, spacious, priv balcony, storage closets, newly-updated kitchen, carpet & paint, gated offstr res parking, 1 bdrm/1 bth, $900, 2 bdrm/1 bth $1,100, Evan, X6784, 525-7655
- SAN PABLO, share 4 bdrm/2 bth home w/ 2 professionals, 1 & 2 rooms avail, spacious & comf, $500mo, incl util & dish, 1 parking/storage space can be provided for add’l price, Carmen, (650) 743-0038, Ccausso @yahoo.com
- SAN PABLO, 2 bdrms in 4 bdrm/2 bth house, share w/ female, $750/mo util incl, avail 10/01, Liz, X2724, cell, 685-0005, Lbear2500@aol.com
- MISC ITEMS FOR SALE
- ROOFTOP CARGO CARRIER, x-large, weatherproof, 50x36x20, fits all vehicles, $40, Karl, X6129
- VACATION
- OREGON, coast home close to prestigious Bandon Dunes Golf Resort, 3 bdrm/2.5 bth, sleeps 8, ocean views, sunsets, $210-$250/ night, 2 night min, Daniel, (707) 246-0386
- PARIS, FRANCE, nr Eiffel Tower, furn, eleg & sunny 2 bdrm/1 bth apt, avail yr-round by wk/mo, close to stores/rest/trans, 848-1830
- LAKE TAHOE house, 3 bdrm/2-1/2 bth, fenced yrd, quiet, sunny, great views of water & mountains, $195/night, 2 night min, Bob, (925) 376-2211
Flea Market Policy
Ads are accepted only from Berkeley Lab employees, retirees, and onsite DOE personnel. Only items of your own personal property may be offered for sale.
Submissions must include name, affiliation, extension, and home phone. Ads must be submitted in writing (e-mail: fleamarket@lbl.gov, fax: X6641), or mailed/delivered to Bldg. 65.
Ads run one issue only unless resubmitted, and are repeated only as space permits. Ads must be submitted in writing and be no more than 50 words in length. The submission deadline for the April 30 issue is Friday, April 23.
People, AWARDS & HONORS

Lab Director Steven Chu Named to
NVIDIA Board
NVIDIA Corporation, a market leader in visual computing
technology, has recently announced the appointment of Berkeley Lab
Director Steven Chu to its board of directors. “The NVIDIA Board is
gaining a world renowned physicist and visionary scientific leader,” said
Jen-Hsun Huang, president and CEO of NVIDIA. Headquartered in Santa Clara,
NVIDIA specializes in graphics and communications processors for PCs and
games.

Lithgow is New Lab Patent Manager
Timothy
Lithgow, whose experience includes both private-sector businesses
and prominent California law firms, became Berkeley Lab’s
new patent manager last month. A patent attorney, Lithgow graduated
from UC Berkeley’s Boalt Hall Law School in 1991.

Nikaido Recieves Freedom Award
Hiroshi Nikaido,
a Berkeley Lab and UC Berkeley biochemist specializing in antibiotic
resistance, has received the 14th annual “Freedom
to Discover Award for Distinguished Achievement in Infectious
Disease Research” from Bristol-Myers Squibb. The award includes a
cash prize of $50,000.
Nikaido was recognized for “groundbreaking
contributions” that
have helped scientists better understand why some bacteria are
resistant to antibiotics. His work has led to the design of more
effective antibiotics.
A native of Tokyo, Nikaido holds degrees from the Keio University
School of Medicine in Tokyo.

Wray Wins ASHRAE Award
The American Society of
Heating, Refrigerating and Air-Conditioning Engineers (ASHRAE)
recently awarded Craig Wray, a mechanical engineer in Berkeley
Lab’s Environmental Energy Technologies Division,
the Distinguished Service Award. The award honors ASHRAE members
who have given freely of their time and talent to the Society.
IN MEMORIAM

Frank McClure
Frank McClure, a leading Bay Area earthquake engineer and researcher
and senior staff scientist at Berkeley Lab before his retirement in 1992,
died on July 16 at his home in Orinda. He was 80 years old.
McClure served as head of the Lab’s Civil/Structural Section of Plant
Engineering from 1978 to 1989 and played a key role in the University
of California Seismic Safety program. A native San Franciscan, Mr. McClure
traveled the world investigating earthquake-damaged buildings to
learn how to better protect structures. In 1990 he received the U.S. Department
of Energy’s Distinguished Associate Award in recognition of his 46
years of leadership in his field.

Paul Silverman
Paul H. Silverman, a biologist and ethicist who held leadership
positions throughout academia, died July 15 at UC Irvine’s Medical
Center from complications from a heart attack. He was 79.
Silverman served as a senior scientist at Berkeley Lab from 1984
through 1990. In 1987 he helped create a first-of-its-kind human genome
center with UC Berkeley,
and In 1993 joined the faculty of
UC Irvine.
Throughout his career, Silverman spoke out about his concerns regarding the social implications of genetics, as well as about the need to pursue stem-cell research.